UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): March 19, 2026
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification Number) | ||||||
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (857 ) 264-4280
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
The | ||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01. Regulation FD Disclosure.
On March 19, 2026, Rhythm Pharmaceuticals, Inc. (“Rhythm”) issued a press release and published a presentation announcing that the U.S. Food and Drug Administration (FDA) has approved an expanded indication for IMCIVREE® (setmelanotide) to treat patients living with acquired hypothalamic obesity (HO), which is summarized under Item 8.01 below. The presentation is available in the “Events and Presentations” portion of the Company’s website at ir.rhythmtx.com. A copy of the press release and presentation are furnished as Exhibits 99.1 and 99.2, respectively, to this Current Report on Form 8-K.
The information contained in Item 7.01 of this Current Report on Form 8-K (including Exhibits 99.1 and 99.2 attached hereto) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), or the Exchange Act, except as expressly provided by specific reference in such a filing.
Item 8.01. Other Events.
On March 19, 2026, Rhythm announced that the U.S. Food and Drug Administration (FDA) has approved an expanded indication for IMCIVREE® (setmelanotide) to treat patients living with acquired hypothalamic obesity (HO). With this expansion, IMCIVREE is indicated to reduce excess body weight and maintain reduction long term in adults and pediatric patients aged 4 years and older with acquired HO.
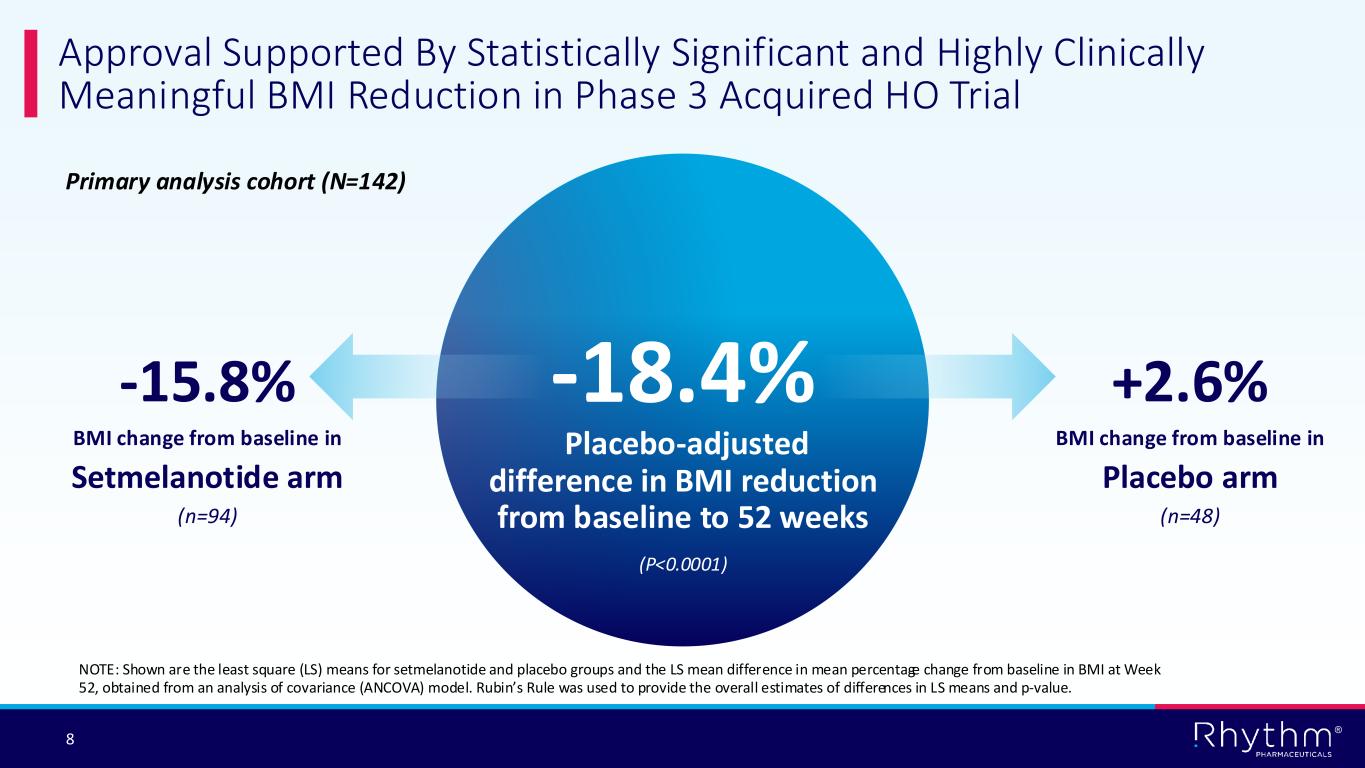
The approval is supported by the positive pivotal Phase 3 TRANSCEND trial of setmelanotide in 142 patients with acquired HO. The global study met its primary endpoint, with a statistically significant -18.4% placebo-adjusted reduction in body mass index (BMI). For the primary endpoint of mean BMI change from baseline, study participants on setmelanotide therapy (n=94) achieved a -15.8% reduction compared with a +2.6% increase among patients on placebo (n=48) at 52 weeks (p<0.0001). Setmelanotide was generally well tolerated in the Phase 3 trial. The most common treatment-emergent adverse events (affecting >20% of participants) were skin hyperpigmentation, nausea, vomiting and headache.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
The following Exhibits 99.1 and 99.2 shall be deemed to be furnished and not filed.
| Exhibit No. | Description | |||||||
| 99.1 | ||||||||
| 99.2 | ||||||||
| 104 | Cover Page Interactive Data File (embedded within the inline XBRL document) | |||||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| RHYTHM PHARMACEUTICALS, INC. | ||||||||
| Date: March 20, 2026 | By: | /s/ Hunter Smith | ||||||
| Hunter Smith | ||||||||
| Chief Financial Officer | ||||||||
EXHIBIT 99.1
Rhythm Pharmaceuticals Announces FDA Approval of IMCIVREE® (setmelanotide) for Patients with Acquired Hypothalamic Obesity
-- First and only FDA-approved therapy for the treatment of acquired hypothalamic obesity, a rare disease characterized by accelerated and sustained weight gain caused by hypothalamic injury or dysfunction –
-- Indicated to reduce excess body weight and maintain reduction long term in adults and pediatric patients aged 4 years and older with acquired hypothalamic obesity --
-- Approval based on -18.4% placebo-adjusted BMI reduction achieved by setmelanotide in global Phase 3 TRANSCEND trial [N=142] --
-- Company to host conference call today at 7:00 p.m. ET --
BOSTON, March 19, 2026 – Rhythm Pharmaceuticals, Inc. (Nasdaq: RYTM), a global commercial-stage biopharmaceutical company focused on transforming the lives of patients living with rare neuroendocrine diseases, today announced that the U.S. Food and Drug Administration (FDA) has approved an expanded indication for IMCIVREE® (setmelanotide) to treat patients living with acquired hypothalamic obesity (HO).
Acquired HO is a rare disease characterized by accelerated and sustained weight gain caused by an injury to the hypothalamus or hypothalamic dysfunction. With this label expansion, IMCIVREE is indicated to reduce excess body weight and maintain reduction long term in adults and pediatric patients aged 4 years and older with acquired HO.
“IMCIVREE is now the first and only FDA-approved therapy for acquired HO, offering a targeted approach that addresses the underlying biology of this disease and meets a critical unmet need for patients who previously had no treatment options,” said David Meeker, M.D., Chairman, Chief Executive Officer and President of Rhythm. “This is a transformative milestone for Rhythm and reinforces our commitment to bringing meaningful therapies to patients living with rare MC4R pathway diseases.”
The MC4R pathway is responsible for controlling physiological functions such as energy expenditure, hunger, and weight regulation. Acquired HO most frequently follows tumors and their treatment or other hypothalamic injury or dysfunction. Based on analysis of the literature, tumor registries and claims data, Rhythm estimates there are approximately 10,000 people living with acquired HO in the U.S.
“Having a therapy for individuals and families affected by acquired hypothalamic obesity has the potential to be transformational,” said Amy Wood, Executive Director and Founder of the Raymond A. Wood Foundation. “We’ve seen firsthand the devastating impact acquired hypothalamic obesity has on patients’ and families’ lives, including relentless hunger and accelerated and sustained weight gain. IMCIVREE offers hope and a path forward for thousands of patients who have long been without options.”
The approval is supported by the positive pivotal Phase 3 TRANSCEND trial of setmelanotide in 142 patients with acquired HO. The global study met its primary endpoint, with a statistically significant -18.4% placebo-adjusted reduction in body mass index (BMI). For the primary endpoint of mean BMI change from baseline, study participants on setmelanotide therapy
(n=94) achieved a -15.8% reduction compared with a +2.6% increase among patients on placebo (n=48) at 52 weeks (p<0.0001). Setmelanotide was generally well tolerated in the Phase 3 trial. The most common adverse events (affecting >20% of participants) were skin hyperpigmentation, nausea, vomiting and headache.
“Setmelanotide has shown effectiveness in targeting the underlying biology of acquired HO,” said Ashley Shoemaker, M.D., MSCI, Associate Professor of Pediatrics, Pediatric Endocrinology at Vanderbilt Health. “Patients treated with setmelanotide experienced meaningful reductions in BMI and hunger, demonstrating the therapy’s ability to deliver clinically significant outcomes in both children and adult patients. Acquired HO is a severe disease that requires early and proactive management. With the availability of IMCIVREE, physicians can offer a targeted therapy.”
IMCIVREE® (setmelanotide) is also approved in the U.S. and Europe in adult and pediatric patients aged 2 years and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS) or Pro-opiomelanocortin (POMC), proprotein convertase subtilisin/kexin type 1 (PCSK1), or leptin receptor (LEPR) deficiency.
Rhythm is committed to supporting access for patients to its medicines and IMCIVREE® (setmelanotide) will be available for patients in the U.S. immediately. Rhythm InTune provides personalized, ongoing educational support for individuals living with certain rare forms of obesity. The program is designed for patients seeking education for themselves and their health care providers, assistance with insurance navigation at treatment initiation, injection support, and guidance on what to expect throughout treatment. For more information, contact patientsupport@rhythmtx.com.
Conference Call Information
Rhythm will host a live conference call and webcast at 5:00 p.m. ET today to discuss the FDA approval of IMCIVREE for patients with acquired HO. To access the live conference call, participants may register here.
Rhythm will host a live conference call and webcast at 5:00 p.m. ET today to discuss the FDA approval of IMCIVREE for patients with acquired HO. To access the live conference call, participants may register here.
A live webcast of the event will be available under "Events and Presentations" in the Investor Relations section of the Rhythm Pharmaceuticals website at https://ir.rhythmtx.com/. The archived webcast will be available on Rhythm Pharmaceuticals’ website approximately two hours after the event and will be available for 30 days following the event.
About Acquired Hypothalamic Obesity
Acquired hypothalamic obesity is a rare disease characterized by accelerated and sustained weight gain caused by an injury to the hypothalamus. Hypothalamic injury may lead to decreased alpha-melanocyte-stimulating hormone (α-MSH) production and impairment of MC4R pathway signaling. The MC4R pathway is responsible for regulating energy balance and body weight. Acquired hypothalamic obesity most frequently follows the growth or treatment of craniopharyngioma, astrocytoma or other hypothalamic-pituitary tumors. Additional causes of injury may include traumatic brain injury, stroke or inflammation. Due to impairment of the MC4R pathway, patients experience accelerated and sustained weight gain, often accompanied by hyperphagia and/or decreased energy expenditure. Acquired hypothalamic obesity can occur as early as six months following hypothalamic injury. Rhythm estimates there are approximately 10,000 people living with acquired HO in the U.S.
About Rhythm Pharmaceuticals
Rhythm is a commercial-stage biopharmaceutical company committed to transforming the lives of patients and their families living with rare neuroendocrine diseases. Rhythm’s lead asset, IMCIVREE® (setmelanotide), an MC4R agonist designed to treat hyperphagia and severe obesity, is approved by the U.S. Food and Drug Administration (FDA) to reduce excess body weight and maintain reduction long term in adults and pediatric patients aged 4 years and older with acquired hypothalamic obesity, adult and pediatric patients 2 years of age and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS) or genetically confirmed pro-opiomelanocortin (POMC), including proprotein convertase subtilisin/kexin type 1 (PCSK1), deficiency or leptin receptor (LEPR) deficiency. Both the European Commission (EC) and the UK’s Medicines & Healthcare Products Regulatory Agency (MHRA) have authorized setmelanotide for the treatment of obesity and the control of hunger associated with genetically confirmed BBS or genetically confirmed loss-of-function biallelic POMC, including PCSK1, deficiency or biallelic LEPR deficiency in adults and children 2 years of age and above. Additionally, Rhythm is advancing a broad clinical development program for setmelanotide in other rare diseases, as well as investigational MC4R agonists bivamelagon and RM-718, and a preclinical suite of small molecules for the treatment of congenital hyperinsulinism. Rhythm’s headquarters is in Boston, MA.
Setmelanotide Indication
In the United States, setmelanotide is indicated to reduce excess body weight and maintain weight reduction long term in adults and pediatric patients aged 4 years and older with acquired hypothalamic obesity, in adult and pediatric patients aged 2 years and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS) or Pro-opiomelanocortin (POMC), proprotein convertase subtilisin/kexin type 1 (PCSK1), or leptin receptor (LEPR) deficiency confirmed by genetic testing demonstrating variants in POMC, PCSK1, or LEPR genes that are interpreted as pathogenic, likely pathogenic, or of uncertain significance (VUS).
In the European Union and the United Kingdom, setmelanotide is indicated for the treatment of obesity and the control of hunger associated with genetically confirmed BBS or loss-of-function biallelic POMC, including PCSK1, deficiency or biallelic LEPR deficiency in adults and children 2 years of age and above. In the European Union and the United Kingdom, setmelanotide should be prescribed and supervised by a physician with expertise in obesity with underlying genetic etiology.
Limitations of Use
Setmelanotide is not indicated for the treatment of patients with the following conditions as setmelanotide would not be expected to be effective:
•Obesity due to suspected POMC, PCSK1, or LEPR deficiency with POMC, PCSK1, or LEPR variants classified as benign or likely benign
•Other types of obesity not related to acquired HO, BBS, or POMC, PCSK1 or LEPR deficiency, including obesity associated with other genetic syndromes and general (polygenic) obesity.
Important Safety Information
CONTRAINDICATIONS
Prior serious hypersensitivity to setmelanotide or any of the excipients in IMCIVREE. Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported.
WARNINGS AND PRECAUTIONS
Disturbance in Sexual Arousal: Spontaneous penile erections and increased frequency of penile erections in males have occurred. Inform patients that these events may occur and instruct patients who have an erection lasting longer than 4 hours to seek emergency medical attention.
Depression and Suicidal Ideation: Depression and suicidal ideation have occurred. Monitor patients for new onset or worsening depression or suicidal thoughts or behaviors. Consider discontinuing IMCIVREE if patients experience suicidal thoughts or behaviors, or clinically significant or persistent depression symptoms occur.
Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported. If suspected, advise patients to promptly seek medical attention and discontinue IMCIVREE.
Skin Hyperpigmentation, Darkening of Pre-existing Nevi, and Development of New Melanocytic Nevi: Generalized or focal increases in skin pigmentation occurred in the majority of IMCIVREE-treated patients. IMCIVREE may also cause development of new melanocytic nevi or darkening of pre-existing nevi. Perform a full body skin examination prior to initiation and periodically during treatment to monitor pre-existing and new pigmented lesions.
Acute Adrenal Insufficiency with Acquired HO: Patients with acquired HO and secondary adrenal insufficiency reported serious adverse reactions related to acute adrenal insufficiency in 5% of IMCIVREE-treated patients and no placebo-treated patients. In patients with secondary adrenal insufficiency, monitor for clinical signs of acute adrenal insufficiency.
Sodium Imbalance in Patients with Acquired HO and Central Diabetes Insipidus: Patients with acquired HO and concomitant central diabetes insipidus (DI)/arginine vasopressin (AVP) deficiency reported hyponatremia in 6% of IMCIVREE-treated patients and 2% of placebo-treated patients and hypernatremia in 5% of IMCIVREE-treated patients and 4% of placebo-treated patients. Monitor serum sodium levels with changes in fluid intake and hydration status. Adjust the doses of concomitant therapies for DI/AVP deficiency as needed.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥20% in at least 1 indication) included skin hyperpigmentation, injection site reactions, nausea, headache, diarrhea, abdominal pain, vomiting, depression, and spontaneous penile erection.
USE IN SPECIFIC POPULATIONS
Treatment with IMCIVREE is not recommended when breastfeeding. Discontinue IMCIVREE when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
To report SUSPECTED ADVERSE REACTIONS, contact Rhythm Pharmaceuticals at +1 (833) 789-6337 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch. See section 4.8 of the Summary of Product Characteristics for information on reporting suspected adverse reactions in Europe.
Please see the full Prescribing Information for additional Important Safety Information.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the safety, efficacy, potential benefits of, and clinical design or progress of any of our products or product candidates at any dosage or in any indication; the approval and use of IMCIVREE in patients with acquired hypothalamic obesity and its availability for patients; the commercial growth of IMCIVREE; our expectations surrounding potential regulatory submissions, progress, or approvals and timing thereof for any of our product candidates; the estimated market size and addressable population for our drug products, including IMCIVREE for the treatment of hypothalamic obesity in the United States, the European Union and Japan; the future announcement of data from our ongoing clinical trials; the ongoing enrollment in and potential progress or outcomes of our clinical trials; the presentation of the full data from the TRANSCEND study at an upcoming medical meeting; and the content, date and timing of any of the foregoing. Statements using words such as “expect”, “anticipate”, “believe”, “may”, “will” and similar terms are also forward-looking statements. Such statements are subject to numerous risks and uncertainties, including, but not limited to, our ability to enroll patients in clinical trials, the design and outcome of clinical trials, the impact of competition, the ability to achieve or obtain necessary regulatory approvals, risks associated with data analysis and reporting, unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, risks associated with the laws and regulations governing our international operations and the costs of any related compliance programs, our ability to successfully commercialize setmelanotide, our liquidity and expenses, our ability to retain our key employees and consultants, and to attract, retain and motivate qualified personnel, and general economic conditions, and the other important factors, including those discussed under the caption “Risk Factors” in Rhythm’s Annual Report on Form 10-K for the year ended December 31, 2025 and our other filings with the Securities and Exchange Commission. Except as required by law, we undertake no obligations to make any revisions to the forward-looking statements contained in this press release or to update them to reflect events or circumstances occurring after the date of this press release, whether as a result of new information, future developments or otherwise.
Corporate Contact:
David Connolly
Head of Investor Relations and Corporate Communications
Rhythm Pharmaceuticals, Inc.
dconnolly@rhythmtx.com
David Connolly
Head of Investor Relations and Corporate Communications
Rhythm Pharmaceuticals, Inc.
dconnolly@rhythmtx.com
Media Contact:
Layne Cosgrove
Real Chemistry
llitsinger@realchemistry.com
Layne Cosgrove
Real Chemistry
llitsinger@realchemistry.com

© Rhythm® Pharmaceuticals, Inc. All rights reserved. ® March 19, 2026 Rhythm Pharmaceuticals IMCIVREE (Setmelanotide) FDA Approval in Acquired Hypothalamic Obesity EXHIBIT 99.2

® 2 On Today’s Call • David Connolly, Vice President of Investor Relations and Corporate Communications • David Meeker, MD, Chair, President and Chief Executive Officer • Jennifer Lee, Executive Vice President, Head of North America • Hunter Smith, Chief Financial Officer • Alicia Fiscus, Senior Vice President, Head of Global Regulatory Affairs

® 3 This presentation and the accompanying oral presentation contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the safety, efficacy, potential benefits of, and clinical design or progress of any of our products or product candidates at any dosage or in any indication; the approval and use of IMCIVREE in patients with acquired hypothalamic obesity and its availability for patients; the commercial growth of IMCIVREE; our expectations surrounding potential regulatory submissions, progress, or approvals and timing thereof for any of our product candidates, including the Type II variation request to the European Medicines Agency and the anticipated decision by the Committee for Medicinal Products for Human Use to issue an opinion to the European Commission and a potential marketing authorization in the second half of 2026; as well as the Company’s engagement with Japan’s Pharmaceuticals and Medical Devices Agency and plans to seek authorization for setmelanotide to treat acquired hypothalamic obesity in Japan; the estimated market size and addressable population for our drug products, including IMCIVREE for the treatment of hypothalamic obesity in the United States, the European Union and Japan; the future announcement of data from our ongoing clinical trials, including the Japanese cohort of our Phase 3 trial evaluating setmelanotide for patients with acquired hypothalamic obesity, the substudy evaluating setmelanotide for patients with congenital hypothalamic obesity; Part C of the Phase 1 trial evaluating RM-718, and the open-label Phase 2 trial evaluating setmelanotide in patients with Prader-Willi syndrome; the ongoing enrollment in and potential progress or outcomes of our clinical trials; the presentation of the full data from the TRANSCEND study at an upcoming medical meeting; and the content, date and timing of any of the foregoing. Statements using words such as “expect”, “anticipate”, “believe”, “may”, “will” and similar terms are also forward-looking statements. Such statements are subject to numerous risks and uncertainties, including, but not limited to, our ability to enroll patients in clinical trials, the design and outcome of clinical trials, the impact of competition, the ability to achieve or obtain necessary regulatory approvals, risks associated with data analysis and reporting, unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, risks associated with the laws and regulations governing our international operations and the costs of any related compliance programs, our ability to successfully commercialize setmelanotide, our liquidity and expenses, our ability to retain our key employees and consultants, and to attract, retain and motivate qualified personnel, and general economic conditions, and the other important factors, including those discussed under the caption “Risk Factors” in Rhythm’s Annual Report on Form 10-K for the year ended December 31, 2025 and our other filings with the Securities and Exchange Commission. Except as required by law, we undertake no obligations to make any revisions to the forward-looking statements contained in this presentation or to update them to reflect events or circumstances occurring after the date of this presentation and the accompanying oral presentation, whether as a result of new information, future developments or otherwise. Forward-looking Statements

® 4 David Meeker, MD Chair, President and CEO

® 5

® 6 INDICATIONS AND USAGE IMCIVREE is a melanocortin 4 (MC4) receptor agonist indicated to reduce excess body weight and maintain reduction long term in adults and pediatric patients aged: • 4 years and older with acquired hypothalamic obesity (HO). • 2 years and older with Bardet-Biedl syndrome (BBS). • 2 years and older with pro-opiomelanocortin (POMC), proprotein convertase subtilisin/kexin type 1 (PCSK1), or leptin receptor (LEPR) deficiency confirmed by genetic testing demonstrating variants in POMC, PCSK1, or LEPR genes that are interpreted as pathogenic, likely pathogenic, or of uncertain significance (VUS). Limitations of Use: IMCIVREE is not indicated for the treatment of patients with the following conditions as IMCIVREE would not be expected to be effective: • Obesity due to suspected POMC, PCSK1, or LEPR deficiency with POMC, PCSK1, or LEPR variants classified as benign or likely benign. • Other types of obesity not related to acquired HO, BBS, or POMC, PCSK1 or LEPR deficiency, including obesity associated with other genetic syndromes and general (polygenic) obesity. IMCIVREE: First and Only Therapy Approved for Acquired HO NOTE: See the full U.S. FDA-approved Package Insert for complete information.

® 7 DOSAGE AND ADMINISTRATION Recommended starting dosage injected subcutaneously for Adults and Pediatric patients aged 6 years and older with Acquired HO: • 0.5 mg (0.05 mL) once daily for 2 weeks. • 1 mg (0.1 mL) once daily for weeks 3-4. • 2 mg (0.2 mL) once daily for weeks 5-6. • 3 mg (0.3 mL) once daily for weeks 7 and onward. ADVERSE REACTIONS • Most common adverse reactions (incidence ≥20% in at least 1 indication) included skin hyperpigmentation, injection site reactions, nausea, headache, diarrhea, abdominal pain, vomiting, depression, and spontaneous penile erection. WARNING AND PRECAUTIONS • Disturbance in Sexual Arousal • Depression and Suicidal Ideation • Hypersensitivity Reactions • Skin Hyperpigmentation, Darkening of Pre-Existing Nevi, and Development of New Melanocytic Nevi • Acute Adrenal Insufficiency in Patients with Acquired HO: Monitor patients for signs of acute adrenal insufficiency • Sodium Imbalance in Patients with Acquired HO and Central Diabetes Insipidus Highlights of U.S. Prescribing Information NOTE: See the full U.S. FDA-approved Package Insert for complete information.
® 8 Approval Supported By Statistically Significant and Highly Clinically Meaningful BMI Reduction in Phase 3 Acquired HO Trial Primary analysis cohort (N=142) -18.4% Placebo-adjusted difference in BMI reduction from baseline to 52 weeks (P<0.0001) -15.8% BMI change from baseline in Setmelanotide arm (n=94) +2.6% BMI change from baseline in Placebo arm (n=48) NOTE: Shown are the least square (LS) means for setmelanotide and placebo groups and the LS mean difference in mean percentage change from baseline in BMI at Week 52, obtained from an analysis of covariance (ANCOVA) model. Rubin’s Rule was used to provide the overall estimates of differences in LS means and p-value.
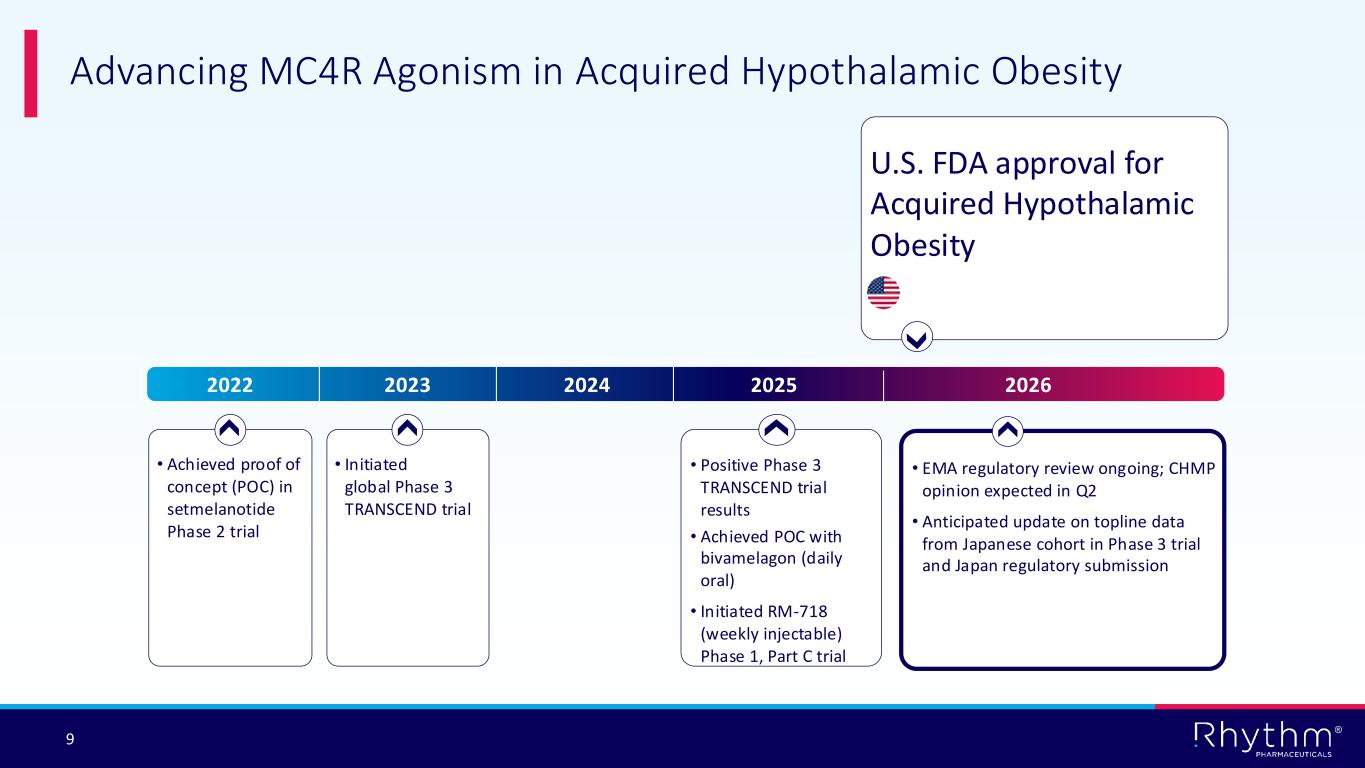
® 9 Advancing MC4R Agonism in Acquired Hypothalamic Obesity 2022 2023 2024 2025 2026 • Achieved proof of concept (POC) in setmelanotide Phase 2 trial • Initiated global Phase 3 TRANSCEND trial • Positive Phase 3 TRANSCEND trial results • Achieved POC with bivamelagon (daily oral) • Initiated RM-718 (weekly injectable) Phase 1, Part C trial • EMA regulatory review ongoing; CHMP opinion expected in Q2 • Anticipated update on topline data from Japanese cohort in Phase 3 trial and Japan regulatory submission U.S. FDA approval for Acquired Hypothalamic Obesity

® 10 Jennifer Lee EVP, Head of North America
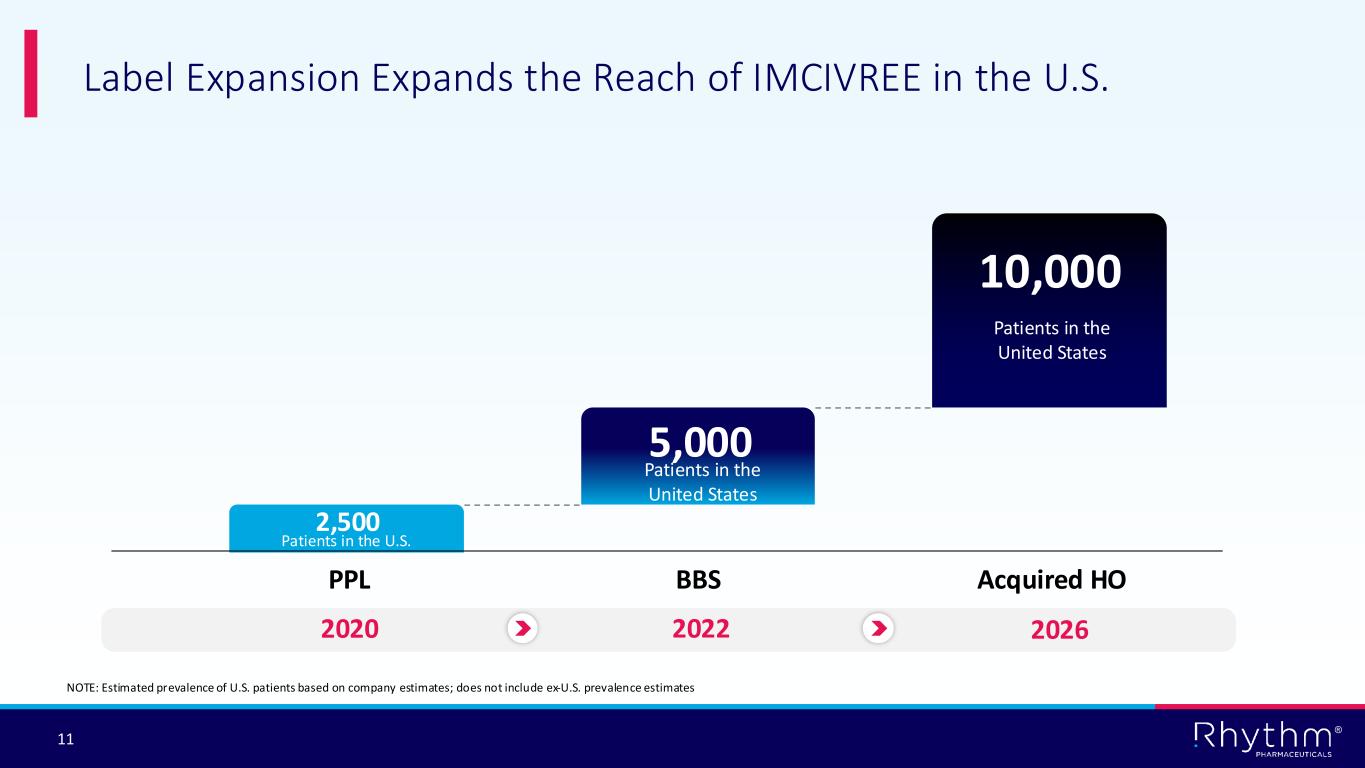
® 11 Label Expansion Expands the Reach of IMCIVREE in the U.S. NOTE: Estimated prevalence of U.S. patients based on company estimates; does not include ex-U.S. prevalence estimates PPL BBS Acquired HO 2020 2022 2026 2,500 5,000 10,000 Patients in the United States Patients in the United States Patients in the U.S.
® 12 Experienced Rare Disease Team in Place to Support Launch Territory managers in the field Access team supporting reimbursement
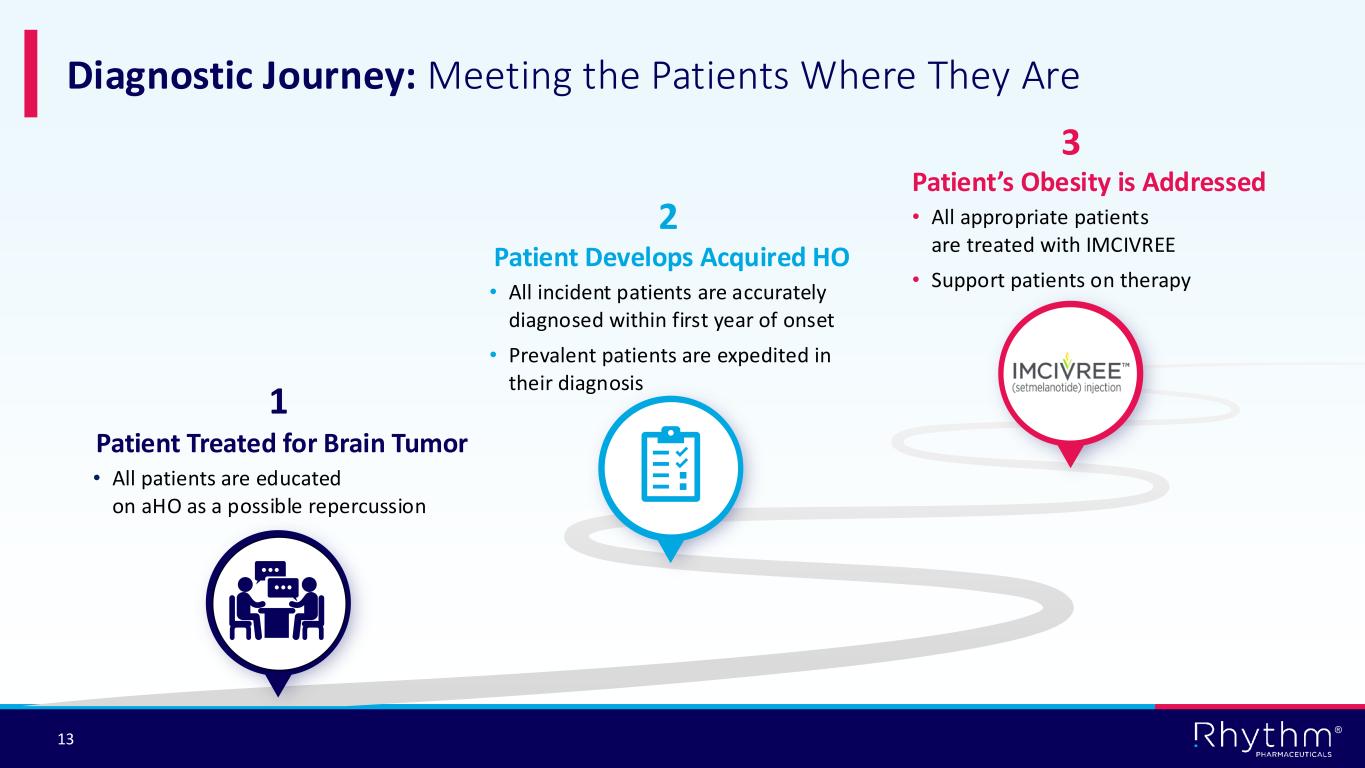
® 13 Diagnostic Journey: Meeting the Patients Where They Are Patient Treated for Brain Tumor • All patients are educated on aHO as a possible repercussion 1 Patient Develops Acquired HO • All incident patients are accurately diagnosed within first year of onset • Prevalent patients are expedited in their diagnosis 2 Patient’s Obesity is Addressed • All appropriate patients are treated with IMCIVREE • Support patients on therapy 3

® 14 Setmelanotide Product Profile Resonates with Endocrinologists In a hypothetical placebo-controlled trial, how compelling would you find 15% BMI reduction vs 0% for placebo? I love seeing that there is improvement in a patient-centered outcome – that being the change in hunger. For me, it’s not all weight-centric all the time – I think we get really focused on that” - Endocrinologist 94% 100% (N=50) extremely compelling or compelling would prescribe setmelanotide to patients with HO
® 15 Transformative Day for HO Community Patients waiting for a therapy First and only approved therapy Ready to launch

® 16 Questions
