UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): March 16, 2026
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification Number) | ||||||
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (857 ) 264-4280
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
The | ||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01. Regulation FD Disclosure.
On March 16, 2026, Rhythm Pharmaceuticals, Inc. (“Rhythm”) issued a press release and published a presentation announcing topline results from its EMANATE trial, which are summarized under Item 8.01 below. The presentation is available in the “Events and Presentations” portion of the Company’s website at ir.rhythmtx.com. A copy of the press release and presentation are furnished as Exhibits 99.1 and 99.2, respectively, to this Current Report on Form 8-K.
The information contained in Item 7.01 of this Current Report on Form 8-K (including Exhibits 99.1 and 99.2 attached hereto) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), or the Exchange Act, except as expressly provided by specific reference in such a filing.
Item 8.01. Other Events.
On March 16, 2026, Rhythm announced the topline results from its EMANATE trial, which are summarized below.
The primary endpoint was the difference in mean percent change in BMI from baseline to Week 52 versus placebo, analyzed in the modified intent-to-treat (ITT) population using prespecified multiple imputation to account for missing values and discontinuations. Topline results include:
•POMC / PCSK1 Hets (N=78): –4.3% placebo-adjusted reduction in BMI (p=0.15);
•LEPR Hets (N=23): –3.6% placebo-adjusted reduction in BMI (p=0.94);
•SRC1 (NCOA1) (N=73): –4.0% placebo-adjusted reduction in BMI (p=0.12); and
•SH2B1 (N=121): –1.7% placebo-adjusted reduction in BMI (p=0.43).
According to post hoc analyses based on last observation carried forward (LOCF) for missing values, setmelanotide achieved statistically significant and clinically meaningful BMI reductions at Week 52 in the modified intent-to-treat patient populations in the POMC/PCSK1 Hets and SRC1 substudies:
•–5.5% least-squares mean difference in BMI in POMC/PCSK1 Hets patients (n=78; p=0.0010); and
•–6.2% least-squares mean difference in BMI in SRC1 (NCOA1) patients (n=73; p<0.0001).
According to post hoc analyses of genetically confirmed patients who reached Week 52 of treatment, setmelanotide achieved statistically significant and clinically meaningful BMI reductions in patients who completed the 52-week trial in the POMC/PCSK1 Hets and SRC1 substudies:
•–9.7% placebo-adjusted reduction in BMI in POMC/PCSK1 Hets patients (n=41; p=0.0002); and
•–8.0% placebo-adjusted reduction in BMI in SRC1 (NCOA1) patients (n=29; p=0.0158).
No new safety signals were observed with setmelanotide in the EMANATE trial, and the safety profile was consistent with prior clinical studies and commercial experience. The most common treatment-emergent adverse events included skin hyperpigmentation, injection site reactions, nausea, vomiting, and headache.
Rhythm plans to continue the analysis of the EMANATE dataset and evaluate potential clinical development paths forward with SRC1 (NCOA1) and POMC with its next-generation MC4R agonists bivamelagon and RM-718. In addition, the Company will continue to evaluate the potential for MC4R agonism in the genes and gene families previously identified through the exploratory Phase 2 DAYBREAK trial, including the SEMA3 family, PHIP, TBX3 or PLXNA family.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
The following Exhibits 99.1 and 99.2 shall be deemed to be furnished and not filed.
| Exhibit No. | Description | |||||||
| 99.1 | ||||||||
| 99.2 | ||||||||
| 104 | Cover Page Interactive Data File (embedded within the inline XBRL document) | |||||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| RHYTHM PHARMACEUTICALS, INC. | ||||||||
| Date: March 16, 2026 | By: | /s/ Hunter Smith | ||||||
| Hunter Smith | ||||||||
| Chief Financial Officer | ||||||||
EXHIBIT 99.1
Rhythm Pharmaceuticals Announces Topline Results from Phase 3 EMANATE Trial
-- Four substudies did not meet pre-specified primary endpoints --
-- Post hoc analyses show setmelanotide achieved statistically significant BMI reductions in patients with obesity due to a heterozygous variant of the POMC/PCSK1 and SRC1 (NCOA1) genes at 52 weeks –
-- Conference call planned for 4:30 p.m. ET today –
BOSTON, March 16, 2026 – Rhythm Pharmaceuticals, Inc. (Nasdaq: RYTM), a global commercial-stage biopharmaceutical company focused on transforming the lives of patients living with rare neuroendocrine diseases, today announced the topline results from its EMANATE trial. The four substudies of this global, Phase 3 trial evaluating setmelanotide did not meet primary endpoints.
“We are grateful to the patients with rare, genetically-driven MC4R pathway diseases and investigators who participated in this trial,” said David Meeker, MD, Chair, President and Chief Executive Officer of Rhythm Pharmaceuticals. “While we are disappointed the EMANATE substudies did not meet their primary endpoint, we are encouraged by compelling signals from additional analyses of the heterozygous POMC/PCSK1 and SRC1 substudies and learnings that sharpen our ability to identify true loss-of-function variants and inform the development of our next-generation MC4R agonists in rare genetically driven obesity indications.”
Dr. Meeker continued, “These patients continue to face a profound unmet medical need, with no approved treatment options that target the underlying biology of their disease. These results provide important insights that support our commitment to advancing targeted therapies for patients with rare genetic obesities.”
EMANATE was a global, randomized, double‑blind, placebo‑controlled Phase 3 trial designed to evaluate the efficacy and safety of setmelanotide in patients with rare, genetically-driven obesities of the MC4R pathway. The study comprised four independent genetic substudies in patients with obesity due to a heterozygous (Hets) variant of the POMC/PCSK1 gene, the LEPR gene, the SRC1 (NCOA1) gene, and the SH2B1 gene with patients randomized 1:1 to receive setmelanotide or placebo for 52 weeks.
EMANATE Topline Results
The primary endpoint was the difference in mean percent change in BMI from baseline to Week 52 versus placebo, analyzed in the modified intent-to-treat (ITT) population using prespecified multiple imputation to account for missing values and discontinuations. Topline results include:
•POMC / PCSK1 Hets (N=78): –4.3% placebo-adjusted reduction in BMI (p=0.15);
•LEPR Hets (N=23): –3.6% placebo-adjusted reduction in BMI (p=0.94);
•SRC1 (NCOA1) (N=73): –4.0% placebo-adjusted reduction in BMI (p=0.12); and
•SH2B1 (N=121): –1.7% placebo-adjusted reduction in BMI (p=0.43).
According to post hoc analyses based on last observation carried forward (LOCF) for missing values, setmelanotide achieved statistically significant and clinically meaningful BMI reductions at Week 52 in the modified intent-to-treat patient populations in the POMC/PCSK1 Hets and SRC1 substudies:
•–5.5% least-squares mean difference in BMI in POMC/PCSK1 Hets patients (n=78; p=0.0010); and
•–6.2% least-squares mean difference in BMI in SRC1 (NCOA1) patients (n=73; p<0.0001).
According to post hoc analyses of genetically confirmed patients who reached Week 52 of treatment, setmelanotide achieved statistically significant and clinically meaningful BMI reductions in patients who completed the 52-week trial in the POMC/PCSK1 Hets and SRC1 substudies:
•–9.7% placebo-adjusted reduction in BMI in POMC/PCSK1 Hets patients (n=41; p=0.0002); and
•–8.0% placebo-adjusted reduction in BMI in SRC1 (NCOA1) patients (n=29; p=0.0158).
No new safety signals were observed with setmelanotide in the EMANATE trial, and the safety profile was consistent with prior clinical studies and commercial experience. The most common treatment-emergent adverse events included skin hyperpigmentation, injection site reactions, nausea, vomiting, and headache.
Rhythm plans to continue the analysis of the EMANATE dataset and evaluate potential clinical development paths forward with SRC1 (NCOA1) and POMC with its next-generation MC4R agonists bivamelagon and RM-718. In addition, the Company will continue to evaluate the potential for MC4R agonism in the genes and gene families previously identified through the exploratory Phase 2 DAYBREAK trial, including the SEMA3 family, PHIP, TBX3 or PLXNA family.
Conference Call Information
Rhythm Pharmaceuticals will host a live conference call and webcast at 4:30 p.m. ET today to discuss this update. Participants may register for the conference call here. It is recommended that participants join the call ten minutes prior to the scheduled start.
A webcast of the call will also be available under "Events and Presentations" in the Investor Relations section of the Rhythm Pharmaceuticals website at https://ir.rhythmtx.com/. The archived webcast will be available on Rhythm Pharmaceuticals’ website approximately two hours after the conference call and will be available for 30 days following the call.
About Rhythm Pharmaceuticals
Rhythm is a commercial-stage biopharmaceutical company committed to transforming the lives of patients and their families living with rare neuroendocrine diseases. Rhythm’s lead asset,
IMCIVREE® (setmelanotide), an MC4R agonist designed to treat hyperphagia and severe obesity, is approved by the U.S. Food and Drug Administration (FDA) to reduce excess body weight and maintain weight reduction long term in adult and pediatric patients 2 years of age and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS) or genetically confirmed pro-opiomelanocortin (POMC), including proprotein convertase subtilisin/kexin type 1 (PCSK1), deficiency or leptin receptor (LEPR) deficiency. Both the European Commission (EC) and the UK’s Medicines & Healthcare Products Regulatory Agency (MHRA) have authorized setmelanotide for the treatment of obesity and the control of hunger associated with genetically confirmed BBS or genetically confirmed loss-of-function biallelic POMC, including PCSK1, deficiency or biallelic LEPR deficiency in adults and children 2 years of age and above. Additionally, Rhythm is advancing a broad clinical development program for setmelanotide in other rare diseases, as well as investigational MC4R agonists bivamelagon and RM-718, and a preclinical suite of small molecules for the treatment of congenital hyperinsulinism. Rhythm’s headquarters is in Boston, MA.
Setmelanotide Indication
In the United States, setmelanotide is indicated to reduce excess body weight and maintain weight reduction long term in adult and pediatric patients aged 2 years and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS) or Pro-opiomelanocortin (POMC), proprotein convertase subtilisin/kexin type 1 (PCSK1), or leptin receptor (LEPR) deficiency as determined by an FDA-approved test demonstrating variants in POMC, PCSK1, or LEPR genes that are interpreted as pathogenic, likely pathogenic, or of uncertain significance (VUS).
In the European Union and the United Kingdom, setmelanotide is indicated for the treatment of obesity and the control of hunger associated with genetically confirmed BBS or loss-of-function biallelic POMC, including PCSK1, deficiency or biallelic LEPR deficiency in adults and children 2 years of age and above. In the European Union and the United Kingdom, setmelanotide should be prescribed and supervised by a physician with expertise in obesity with underlying genetic etiology.
Limitations of Use
Setmelanotide is not indicated for the treatment of patients with the following conditions as setmelanotide would not be expected to be effective:
•Obesity due to suspected POMC, PCSK1, or LEPR deficiency with POMC, PCSK1, or LEPR variants classified as benign or likely benign
•Other types of obesity not related to BBS or POMC, PCSK1, or LEPR deficiency, including obesity associated with other genetic syndromes and general (polygenic) obesity
Contraindication
Prior serious hypersensitivity to setmelanotide or any of the excipients in IMCIVREE. Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported.
WARNINGS AND PRECAUTIONS
Disturbance in Sexual Arousal: Spontaneous penile erections in males and sexual adverse reactions in females have occurred. Inform patients that these events may occur and instruct patients who have an erection lasting longer than 4 hours to seek emergency medical attention.
Depression and Suicidal Ideation: Depression, suicidal ideation and depressed mood have occurred. Monitor patients for new onset or worsening depression or suicidal thoughts or behaviors. Consider discontinuing IMCIVREE if patients experience suicidal thoughts or behaviors, or clinically significant or persistent depression symptoms occur.
Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported. If suspected, advise patients to promptly seek medical attention and discontinue IMCIVREE.
Skin Hyperpigmentation, Darkening of Pre-existing Nevi, and Development of New Melanocytic Nevi: Generalized or focal increases in skin pigmentation, darkening of pre-existing nevi, development of new melanocytic nevi and increase in size of existing melanocytic nevi have occurred. Perform a full body skin examination prior to initiation and periodically during treatment to monitor pre-existing and new pigmented lesions.
Risk of Serious Adverse Reactions Due to Benzyl Alcohol Preservative in Neonates and Low Birth Weight Infants: IMCIVREE is not approved for use in neonates or infants. Serious and fatal adverse reactions including “gasping syndrome” can occur in neonates and low birth weight infants treated with benzyl alcohol preserved drugs.
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥20%) included skin hyperpigmentation, injection site reactions, nausea, headache, diarrhea, abdominal pain, vomiting, depression, and spontaneous penile erection.
USE IN SPECIFIC POPULATIONS
Treatment with IMCIVREE is not recommended when breastfeeding. Discontinue IMCIVREE when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
To report SUSPECTED ADVERSE REACTIONS, contact Rhythm Pharmaceuticals at +1 (833) 789-6337 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See section 4.8 of the Summary of Product Characteristics for information on reporting suspected adverse reactions in Europe.
Please see the full Prescribing Information for additional Important Safety Information.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the potential safety, efficacy, potential benefits of, and clinical design or progress of any of our products or product candidates at any dosage or in any indication; clinical design, enrollment, or progress, and preliminary, interim and final data readouts; potential benefits from our EMANATE topline results, including our ability to identify true loss-of-function variants and develop our next-generation MC4R agonists; potential regulatory submissions, approvals and timing thereof of setmelanotide, bivamelagon, or RM-718; the potential benefits of any of the Company’s products or product candidates for any specific disease indication or at any dosage, including the potential benefits of setmelanotide, bivamelagon or RM-718 for patients with MC4R pathway diseases; expectations surrounding pending and potential regulatory submissions and approvals, including within the United States, the EU and other regions; business strategy and plans, including regarding commercialization of setmelanotide in the United States, the EU and other regions; our participation in upcoming events and presentations; and the timing of any of the foregoing. Statements using words such as “expect”, “anticipate”, “believe”, “may”, “will” and similar terms are also forward-looking statements. Such statements are subject to numerous risks and uncertainties, including, but not limited to, our ability to enroll patients in clinical trials, the design and outcome of clinical trials, the impact of competition, the ability to achieve or obtain necessary regulatory approvals, risks associated with data analysis and reporting, our ability to successfully commercialize setmelanotide, our liquidity and expenses, our ability to retain our key employees and consultants, and to attract, retain and motivate qualified personnel, and general economic conditions, and the other important factors discussed under the caption “Risk Factors” in Rhythm’s Annual Report on Form 10-K for the year ended December 31, 2025 and other filings with the Securities and Exchange Commission. Except as required by law, we undertake no obligations to make any revisions to the forward-looking statements contained in this release or to update them to reflect events or circumstances occurring after the date of this release, whether as a result of new information, future developments or otherwise.
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the potential safety, efficacy, potential benefits of, and clinical design or progress of any of our products or product candidates at any dosage or in any indication; clinical design, enrollment, or progress, and preliminary, interim and final data readouts; potential benefits from our EMANATE topline results, including our ability to identify true loss-of-function variants and develop our next-generation MC4R agonists; potential regulatory submissions, approvals and timing thereof of setmelanotide, bivamelagon, or RM-718; the potential benefits of any of the Company’s products or product candidates for any specific disease indication or at any dosage, including the potential benefits of setmelanotide, bivamelagon or RM-718 for patients with MC4R pathway diseases; expectations surrounding pending and potential regulatory submissions and approvals, including within the United States, the EU and other regions; business strategy and plans, including regarding commercialization of setmelanotide in the United States, the EU and other regions; our participation in upcoming events and presentations; and the timing of any of the foregoing. Statements using words such as “expect”, “anticipate”, “believe”, “may”, “will” and similar terms are also forward-looking statements. Such statements are subject to numerous risks and uncertainties, including, but not limited to, our ability to enroll patients in clinical trials, the design and outcome of clinical trials, the impact of competition, the ability to achieve or obtain necessary regulatory approvals, risks associated with data analysis and reporting, our ability to successfully commercialize setmelanotide, our liquidity and expenses, our ability to retain our key employees and consultants, and to attract, retain and motivate qualified personnel, and general economic conditions, and the other important factors discussed under the caption “Risk Factors” in Rhythm’s Annual Report on Form 10-K for the year ended December 31, 2025 and other filings with the Securities and Exchange Commission. Except as required by law, we undertake no obligations to make any revisions to the forward-looking statements contained in this release or to update them to reflect events or circumstances occurring after the date of this release, whether as a result of new information, future developments or otherwise.
Corporate Contact:
David Connolly
Head of Investor Relations and Corporate Communications
Rhythm Pharmaceuticals, Inc.
857-264-4280
dconnolly@rhythmtx.com
Media Contact:
Layne Cosgrove
Real Chemistry
410-916-1035
llitsinger@realchemistry.com

© Rhythm® Pharmaceuticals, Inc. All rights reserved. ® Rhythm Pharmaceuticals Phase 3 EMANATE Trial Topline Results March 16, 2026 EXHIBIT 99.2

® 2 On Today’s Call • David Connolly, Executive Director of Investor Relations and Corporate Communications • David Meeker, MD, Chair, President and Chief Executive Officer • Alastair Garfield, Chief Scientific Officer • Hunter Smith, Chief Financial Officer

® 3 This presentation and the accompanying oral presentation contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the potential safety, efficacy, potential benefits of, and clinical design or progress of any of our products or product candidates at any dosage or in any indication; clinical design, enrollment, or progress, and preliminary, interim and final data readouts; potential benefits from our EMANATE topline results, including our ability to identify true loss-of-function variants and develop our next-generation MC4R agonists; potential regulatory submissions, approvals and timing thereof of setmelanotide, bivamelagon, or RM-718; the potential benefits of any of the Company’s products or product candidates for any specific disease indication or at any dosage, including the potential benefits of setmelanotide, bivamelagon or RM-718 for patients with MC4R pathway diseases; expectations surrounding pending and potential regulatory submissions and approvals, including within the United States, the EU and other regions; business strategy and plans, including regarding commercialization of setmelanotide in the United States, the EU and other regions; our participation in upcoming events and presentations; and the timing of any of the foregoing. Statements using words such as “expect”, “anticipate”, “believe”, “may”, “will” and similar terms are also forward-looking statements. Such statements are subject to numerous risks and uncertainties, including, but not limited to, our ability to enroll patients in clinical trials, the design and outcome of clinical trials, the impact of competition, the ability to achieve or obtain necessary regulatory approvals, risks associated with data analysis and reporting, our ability to successfully commercialize setmelanotide, our liquidity and expenses, our ability to retain our key employees and consultants, and to attract, retain and motivate qualified personnel, and general economic conditions, and the other important factors discussed under the caption “Risk Factors” in Rhythm’s Annual Report on Form 10-K for the year ended December 31, 2025 and other filings with the Securities and Exchange Commission. Except as required by law, we undertake no obligations to make any revisions to the forward-looking statements contained in this release or to update them to reflect events or circumstances occurring after the date of this release, whether as a result of new information, future developments or otherwise. Forward-Looking Statements

® 4 David Meeker, MD Chair, President and CEO

® 5 Phase 3 EMANATE Topline Results Encouraging signals seen in POMC Hets and SRC1 substudies to inform development of next- generation MC4R agonists • Post hoc LOCF analyses demonstrated setmelanotide achieved statistically significant BMI reduction in POMC Hets and SRC1 substudies Deepened understanding across MC4R pathway genetics and variant classification Placebo effect potentially confounded analysis of intent-to-treat (ITT) population High discontinuation rate across all four substudies negatively affected topline results • Multiple imputation statistical analysis for calculating missing values and discontinued patients confounded primary endpoint analysis
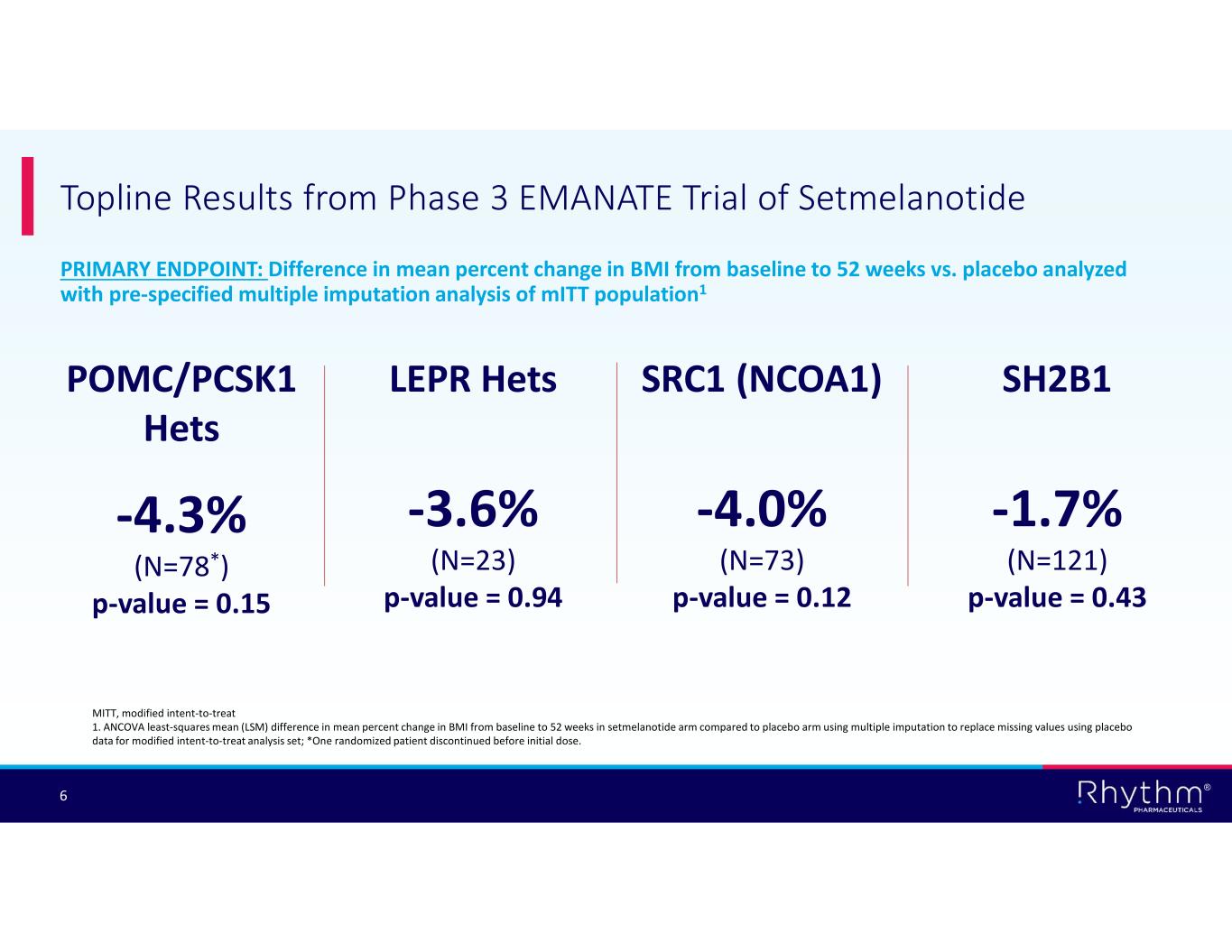
® 6 PRIMARY ENDPOINT: Difference in mean percent change in BMI from baseline to 52 weeks vs. placebo analyzed with pre-specified multiple imputation analysis of mITT population1 Topline Results from Phase 3 EMANATE Trial of Setmelanotide LEPR Hets -3.6% (N=23) p-value = 0.94 SRC1 (NCOA1) -4.0% (N=73) p-value = 0.12 SH2B1 -1.7% (N=121) p-value = 0.43 POMC/PCSK1 Hets -4.3% (N=78*) p-value = 0.15 MITT, modified intent-to-treat 1. ANCOVA least-squares mean (LSM) difference in mean percent change in BMI from baseline to 52 weeks in setmelanotide arm compared to placebo arm using multiple imputation to replace missing values using placebo data for modified intent-to-treat analysis set; *One randomized patient discontinued before initial dose.
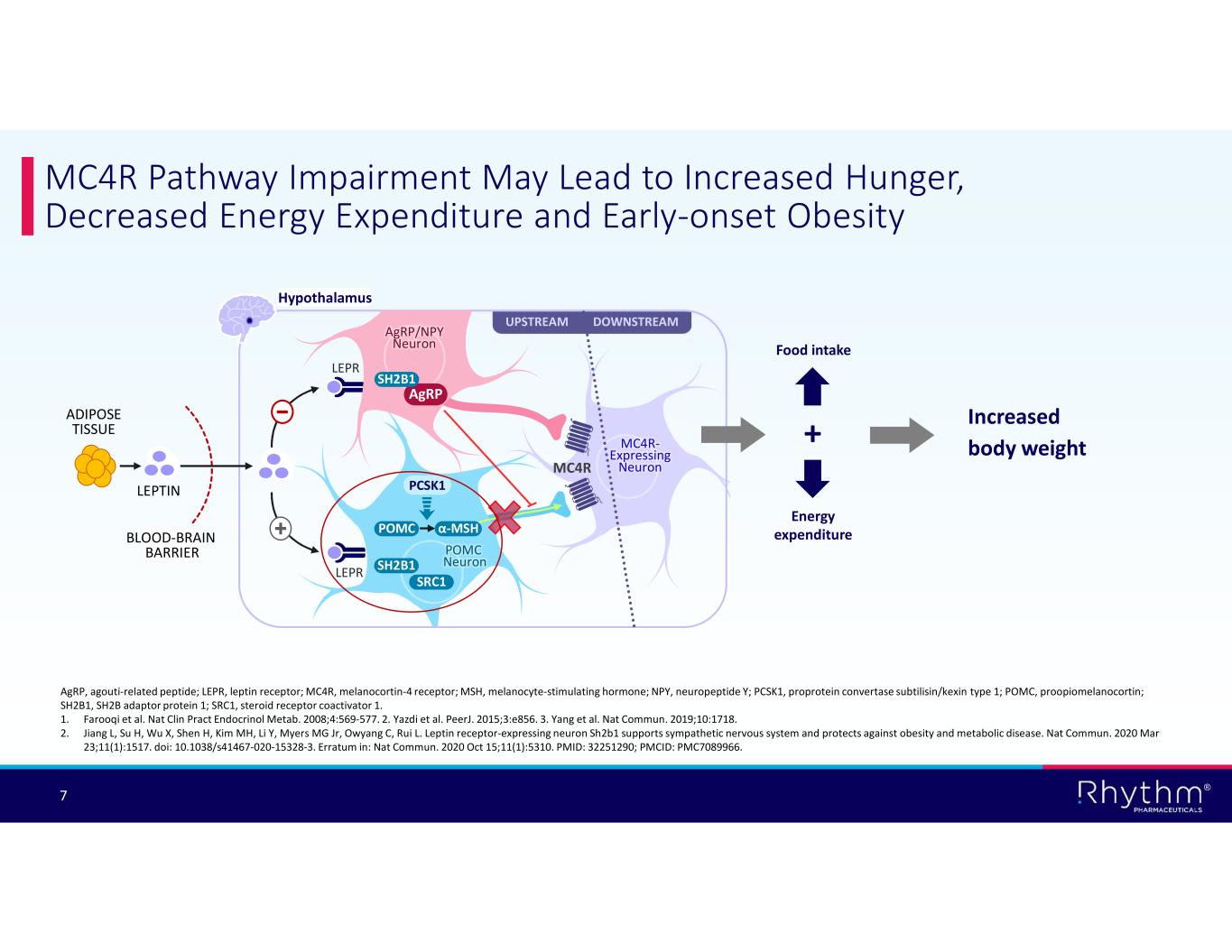
® 7 MC4R Pathway Impairment May Lead to Increased Hunger, Decreased Energy Expenditure and Early-onset Obesity AgRP, agouti-related peptide; LEPR, leptin receptor; MC4R, melanocortin-4 receptor; MSH, melanocyte-stimulating hormone; NPY, neuropeptide Y; PCSK1, proprotein convertase subtilisin/kexin type 1; POMC, proopiomelanocortin; SH2B1, SH2B adaptor protein 1; SRC1, steroid receptor coactivator 1. 1. Farooqi et al. Nat Clin Pract Endocrinol Metab. 2008;4:569-577. 2. Yazdi et al. PeerJ. 2015;3:e856. 3. Yang et al. Nat Commun. 2019;10:1718. 2. Jiang L, Su H, Wu X, Shen H, Kim MH, Li Y, Myers MG Jr, Owyang C, Rui L. Leptin receptor-expressing neuron Sh2b1 supports sympathetic nervous system and protects against obesity and metabolic disease. Nat Commun. 2020 Mar 23;11(1):1517. doi: 10.1038/s41467-020-15328-3. Erratum in: Nat Commun. 2020 Oct 15;11(1):5310. PMID: 32251290; PMCID: PMC7089966. UPSTREAM DOWNSTREAM MC4R- Expressing Neuron PCSK1 MC4R LEPR POMC LEPR POMC Neuron Hypothalamus AgRP/NPY Neuron AgRP α-MSH ADIPOSE TISSUE LEPTIN BLOOD-BRAIN BARRIER SRC1 SH2B1 SH2B1 Increased body weight+ Energy expenditure Food intake
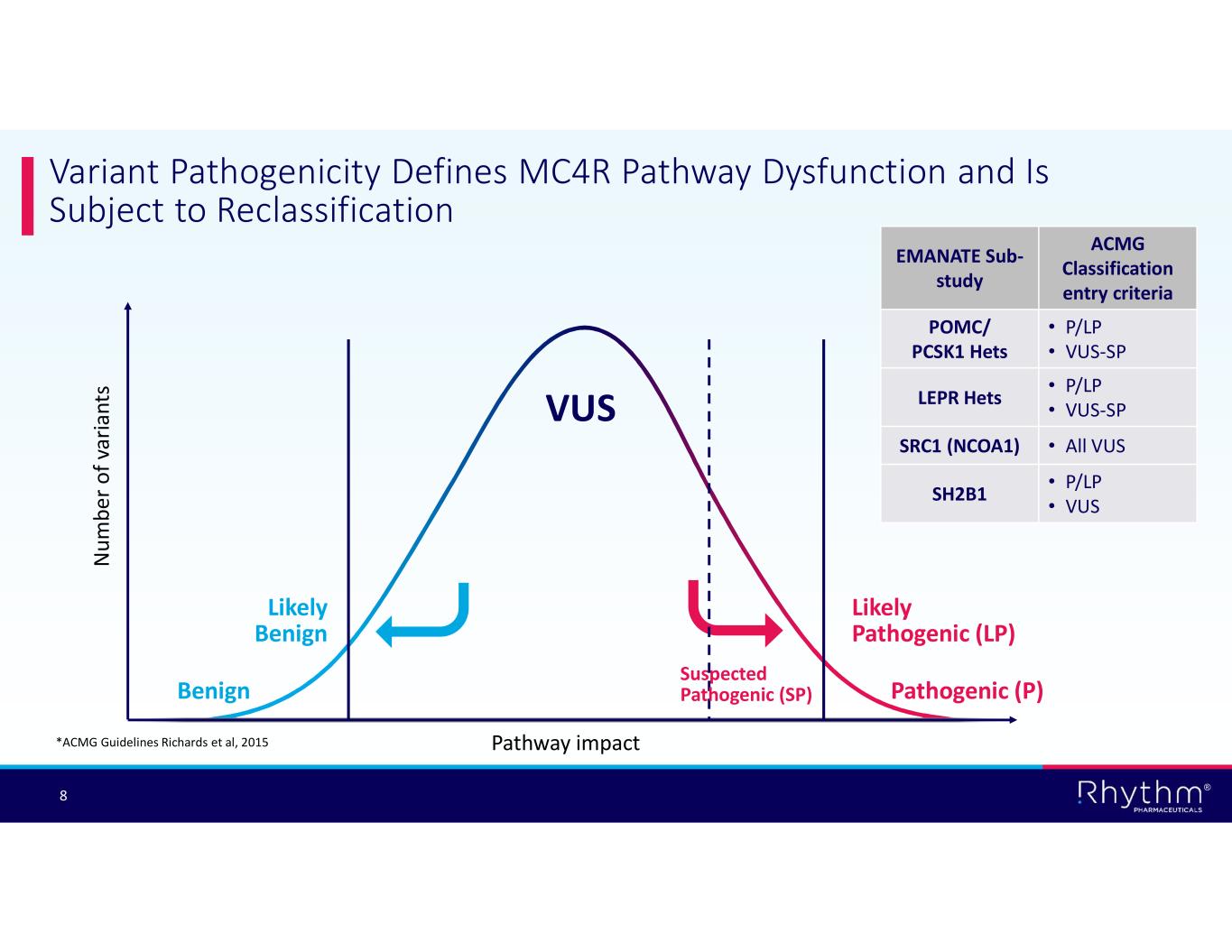
® 8 Variant Pathogenicity Defines MC4R Pathway Dysfunction and Is Subject to Reclassification Pathogenic (P) Likely Pathogenic (LP) VUS Likely Benign Benign N um be r o f v ar ia nt s Pathway impact*ACMG Guidelines Richards et al, 2015 Suspected Pathogenic (SP) ACMG Classification entry criteria EMANATE Sub- study • P/LP • VUS-SP POMC/ PCSK1 Hets • P/LP • VUS-SPLEPR Hets • All VUSSRC1 (NCOA1) • P/LP • VUSSH2B1

® 9 Each sub-study: Patients randomized 1:1 Phase 3 EMANATE Trial Comprised of Four Independent Sub-studies Enrolled patientsGenetic sub-study N=79* POMC/ PCSK1 Hets N=23LEPR Hets N=73SRC1 (NCOA1) N=121SH2B1 Setmelanotide Placebo 52 weeks Open-label extension Primary Endpoint: Difference in mean percent change in BMI from baseline to 52 weeks in setmelanotide arm compared to placebo arm VUS, Variant of uncertain significance *One randomized patient discontinued before initial dose.
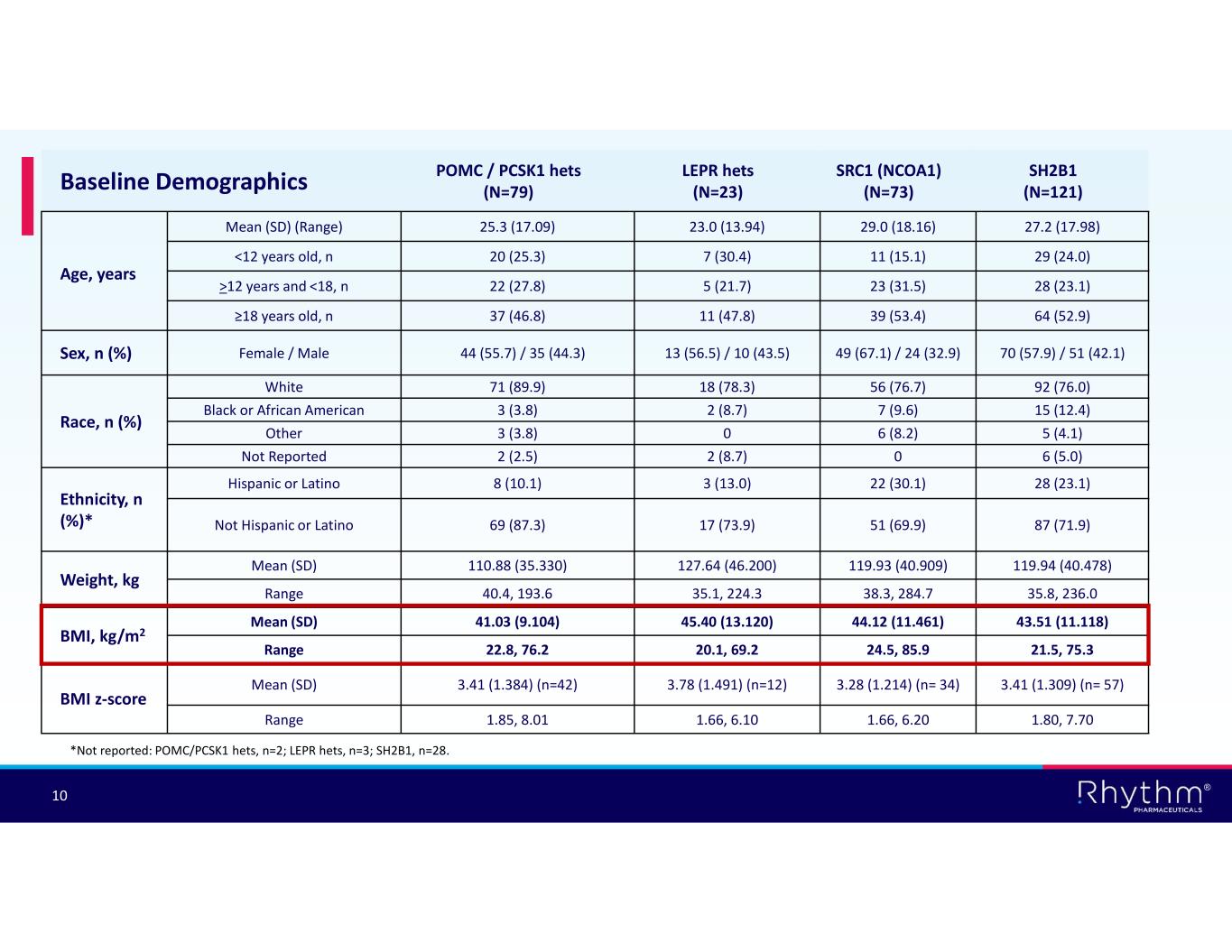
® 10 SH2B1 (N=121) SRC1 (NCOA1) (N=73) LEPR hets (N=23) POMC / PCSK1 hets (N=79)Baseline Demographics 27.2 (17.98)29.0 (18.16)23.0 (13.94)25.3 (17.09) Mean (SD) (Range) Age, years 29 (24.0)11 (15.1)7 (30.4)20 (25.3)<12 years old, n 28 (23.1)23 (31.5)5 (21.7)22 (27.8)>12 years and <18, n 64 (52.9)39 (53.4)11 (47.8)37 (46.8)≥18 years old, n 70 (57.9) / 51 (42.1)49 (67.1) / 24 (32.9)13 (56.5) / 10 (43.5)44 (55.7) / 35 (44.3) Female / MaleSex, n (%) 92 (76.0)56 (76.7)18 (78.3)71 (89.9)White Race, n (%) 15 (12.4)7 (9.6)2 (8.7)3 (3.8)Black or African American 5 (4.1)6 (8.2)03 (3.8)Other 6 (5.0)02 (8.7)2 (2.5)Not Reported 28 (23.1)22 (30.1)3 (13.0)8 (10.1)Hispanic or Latino Ethnicity, n (%)* 87 (71.9)51 (69.9)17 (73.9)69 (87.3)Not Hispanic or Latino 119.94 (40.478)119.93 (40.909)127.64 (46.200)110.88 (35.330)Mean (SD) Weight, kg 35.8, 236.038.3, 284.735.1, 224.340.4, 193.6Range 43.51 (11.118)44.12 (11.461)45.40 (13.120)41.03 (9.104)Mean (SD) BMI, kg/m2 21.5, 75.324.5, 85.920.1, 69.222.8, 76.2Range 3.41 (1.309) (n= 57)3.28 (1.214) (n= 34)3.78 (1.491) (n=12)3.41 (1.384) (n=42)Mean (SD) BMI z-score 1.80, 7.701.66, 6.201.66, 6.101.85, 8.01Range *Not reported: POMC/PCSK1 hets, n=2; LEPR hets, n=3; SH2B1, n=28.
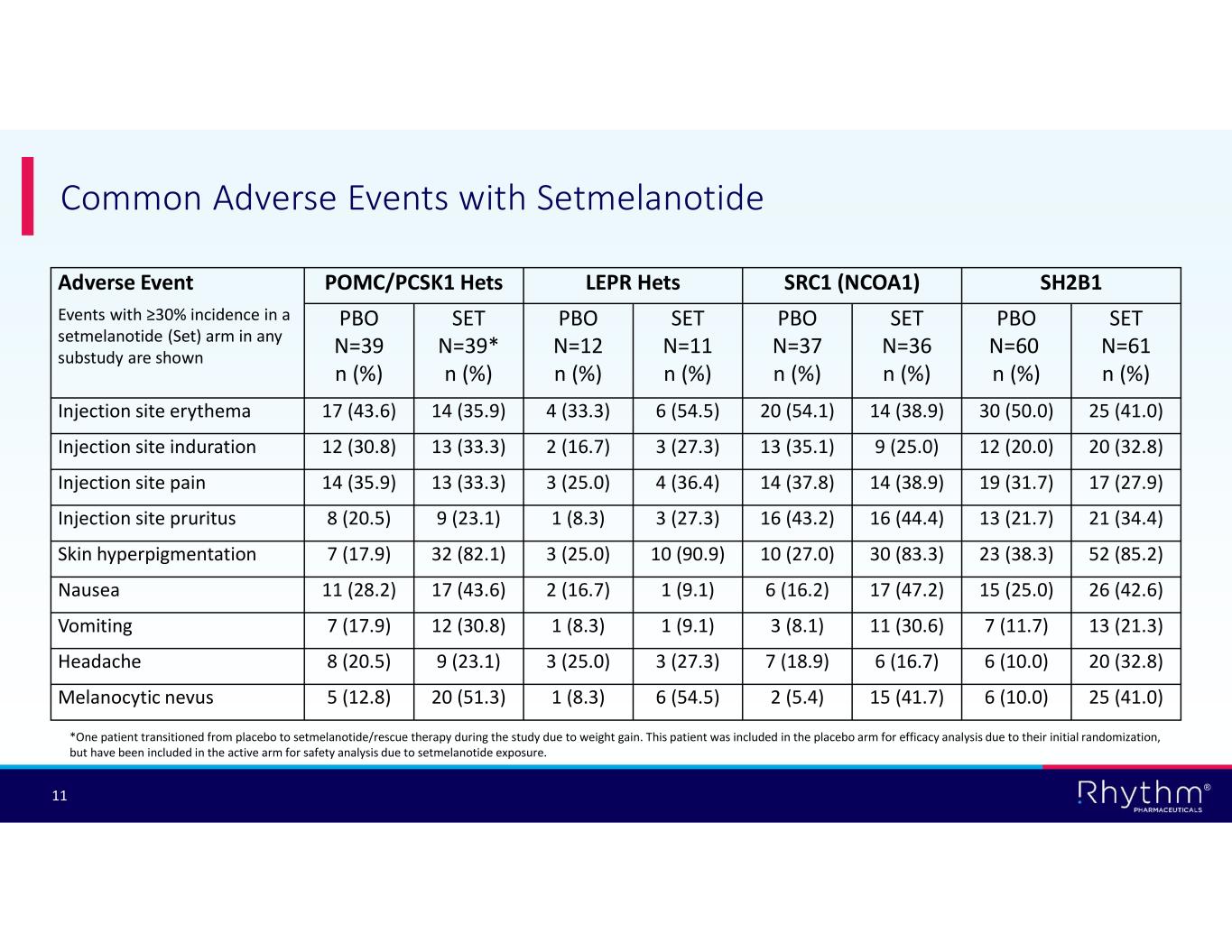
® 11 Common Adverse Events with Setmelanotide SH2B1SRC1 (NCOA1)LEPR HetsPOMC/PCSK1 HetsAdverse Event Events with ≥30% incidence in a setmelanotide (Set) arm in any substudy are shown SET N=61 n (%) PBO N=60 n (%) SET N=36 n (%) PBO N=37 n (%) SET N=11 n (%) PBO N=12 n (%) SET N=39* n (%) PBO N=39 n (%) 25 (41.0)30 (50.0)14 (38.9)20 (54.1)6 (54.5)4 (33.3)14 (35.9)17 (43.6)Injection site erythema 20 (32.8)12 (20.0)9 (25.0)13 (35.1)3 (27.3)2 (16.7)13 (33.3)12 (30.8)Injection site induration 17 (27.9)19 (31.7)14 (38.9)14 (37.8)4 (36.4)3 (25.0)13 (33.3)14 (35.9)Injection site pain 21 (34.4)13 (21.7)16 (44.4)16 (43.2)3 (27.3)1 (8.3)9 (23.1)8 (20.5)Injection site pruritus 52 (85.2)23 (38.3)30 (83.3)10 (27.0)10 (90.9)3 (25.0)32 (82.1)7 (17.9)Skin hyperpigmentation 26 (42.6)15 (25.0)17 (47.2)6 (16.2)1 (9.1)2 (16.7)17 (43.6)11 (28.2)Nausea 13 (21.3)7 (11.7)11 (30.6)3 (8.1)1 (9.1)1 (8.3)12 (30.8)7 (17.9)Vomiting 20 (32.8)6 (10.0)6 (16.7)7 (18.9)3 (27.3)3 (25.0)9 (23.1)8 (20.5)Headache 25 (41.0)6 (10.0)15 (41.7)2 (5.4)6 (54.5)1 (8.3)20 (51.3)5 (12.8)Melanocytic nevus *One patient transitioned from placebo to setmelanotide/rescue therapy during the study due to weight gain. This patient was included in the placebo arm for efficacy analysis due to their initial randomization, but have been included in the active arm for safety analysis due to setmelanotide exposure.
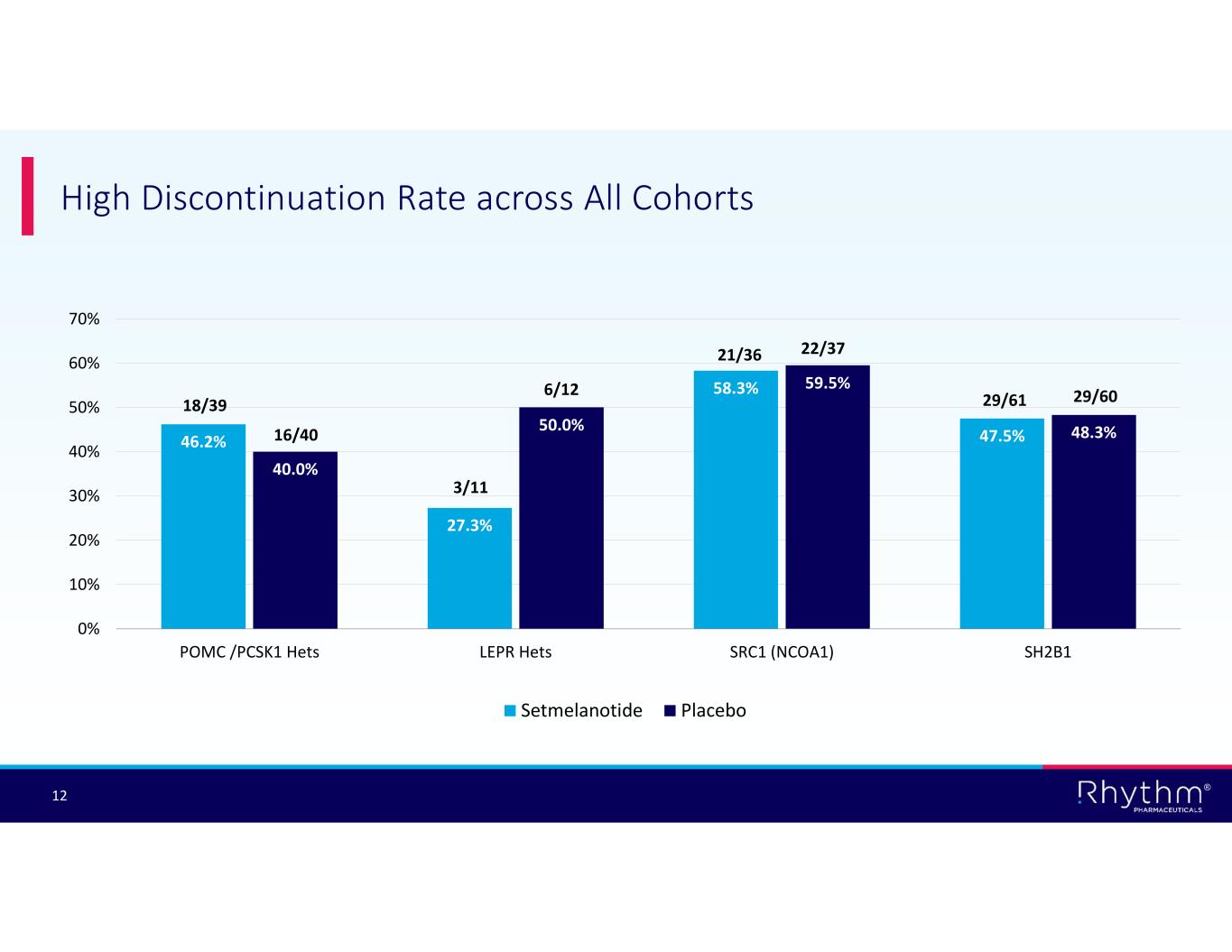
® 12 High Discontinuation Rate across All Cohorts 46.2% 27.3% 58.3% 47.5% 40.0% 50.0% 59.5% 48.3% 0% 10% 20% 30% 40% 50% 60% 70% POMC /PCSK1 Hets LEPR Hets SRC1 (NCOA1) SH2B1 Setmelanotide Placebo 18/39 16/40 3/11 6/12 21/36 22/37 29/61 29/60
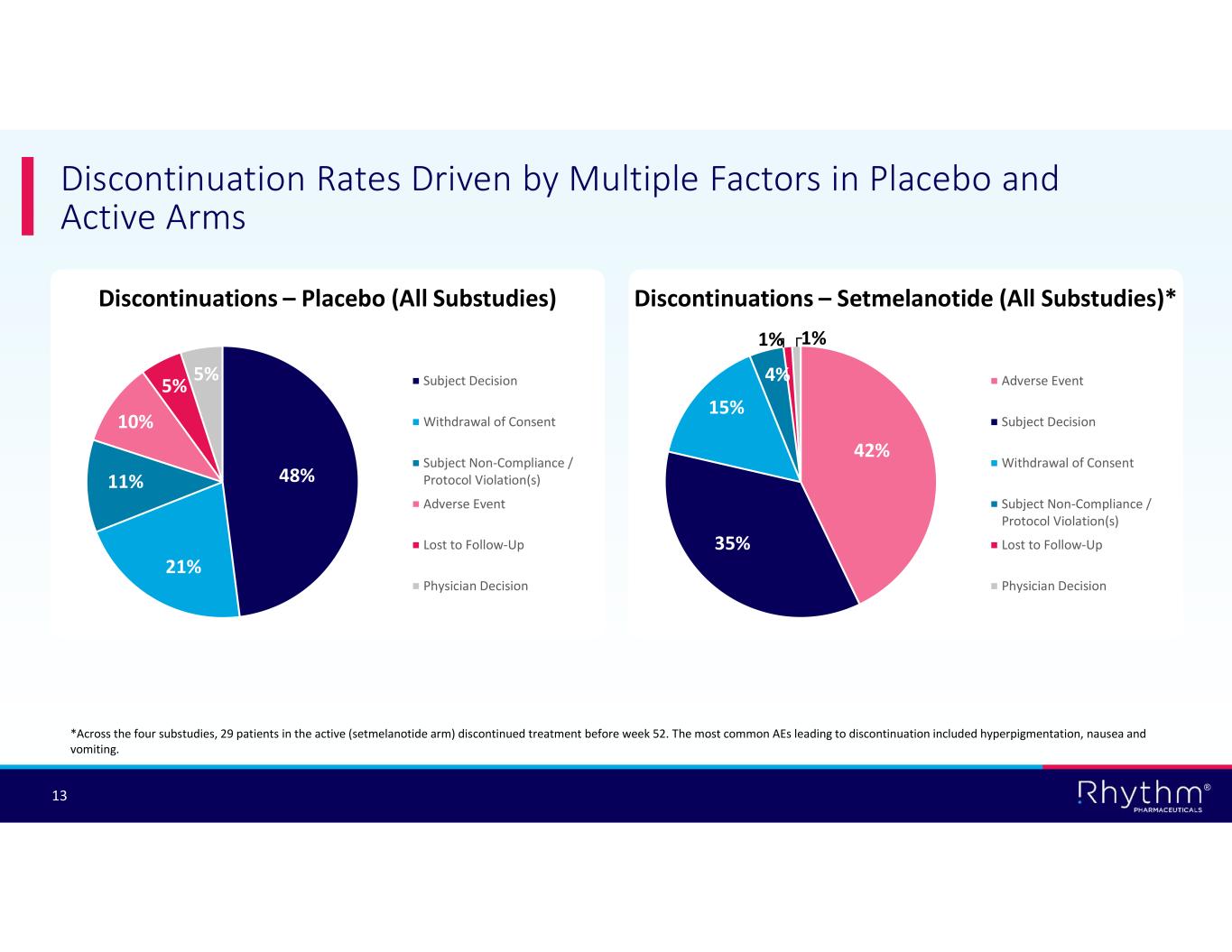
® 13 Discontinuations – Placebo (All Substudies) Discontinuation Rates Driven by Multiple Factors in Placebo and Active Arms 48% 21% 11% 10% 5% 5% Subject Decision Withdrawal of Consent Subject Non-Compliance / Protocol Violation(s) Adverse Event Lost to Follow-Up Physician Decision Discontinuations – Setmelanotide (All Substudies)* 42% 35% 15% 4% 1% 1% Adverse Event Subject Decision Withdrawal of Consent Subject Non-Compliance / Protocol Violation(s) Lost to Follow-Up Physician Decision *Across the four substudies, 29 patients in the active (setmelanotide arm) discontinued treatment before week 52. The most common AEs leading to discontinuation included hyperpigmentation, nausea and vomiting.

® 14 POMC / PCSK1 Hets
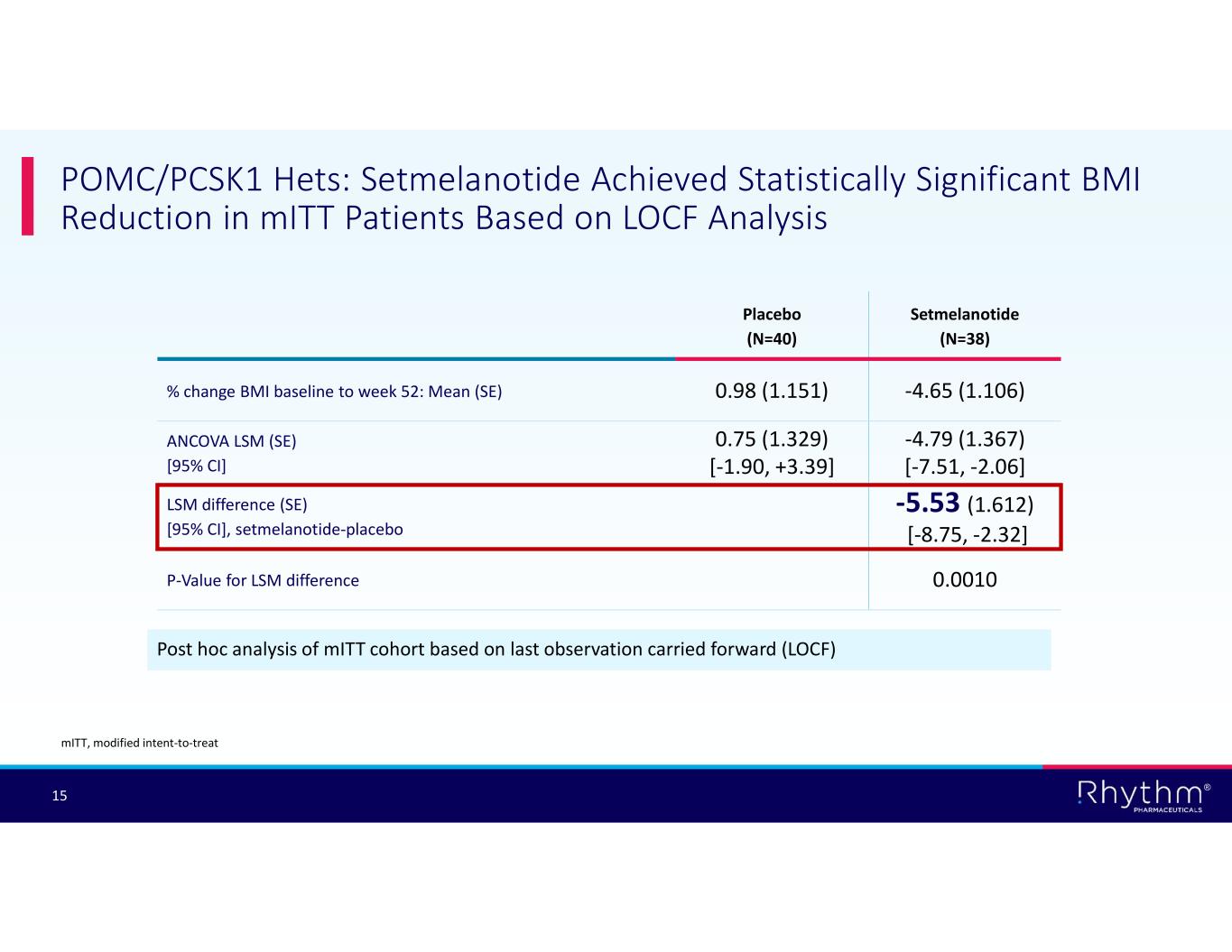
® 15 POMC/PCSK1 Hets: Setmelanotide Achieved Statistically Significant BMI Reduction in mITT Patients Based on LOCF Analysis Post hoc analysis of mITT cohort based on last observation carried forward (LOCF) Setmelanotide (N=38) Placebo (N=40) -4.65 (1.106)0.98 (1.151)% change BMI baseline to week 52: Mean (SE) -4.79 (1.367) [-7.51, -2.06] 0.75 (1.329) [-1.90, +3.39] ANCOVA LSM (SE) [95% CI] -5.53 (1.612) [-8.75, -2.32] LSM difference (SE) [95% CI], setmelanotide-placebo 0.0010P-Value for LSM difference mITT, modified intent-to-treat

® 16 POMC/PCSK1 Hets: Setmelanotide Achieved Statistically Significant BMI Reduction in Genetically Confirmed Patients Based on LOCF Analysis Post hoc analysis of genetically confirmed patients based on last observation carried forward (LOCF) Setmelanotide (N=32) Placebo (N=35) -5.04 (1.171)2.20 (1.290)% change BMI baseline to week 52: Mean (SE) -4.17 (1.511) [-7.19, -1.15] 2.61 (1.628) [-0.64, 5.87] ANCOVA LSM (SE) [95% CI] -6.78 (1.775) [-10.33, -3.23] LSM difference (SE) [95% CI], setmelanotide-placebo 0.0003P-Value for LSM difference
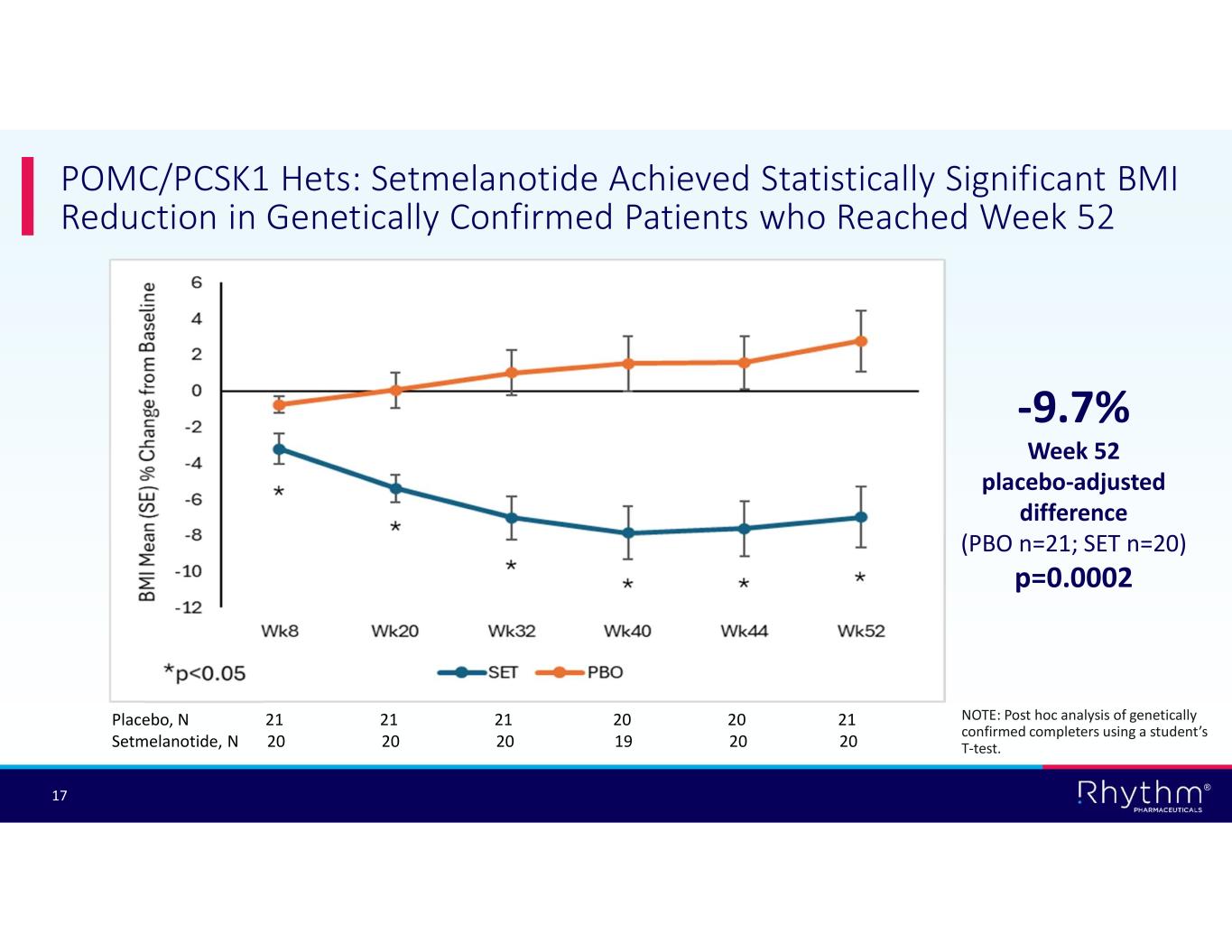
® 17 POMC/PCSK1 Hets: Setmelanotide Achieved Statistically Significant BMI Reduction in Genetically Confirmed Patients who Reached Week 52 -9.7% Week 52 placebo-adjusted difference (PBO n=21; SET n=20) p=0.0002 Placebo, N 21 21 21 20 20 21 Setmelanotide, N 20 20 20 19 20 20 NOTE: Post hoc analysis of genetically confirmed completers using a student’s T-test.

® 18 LEPR Hets

® 19 Highlights from the LEPR Hets Substudy *Patient with -7.2% BMI reduction at Week 28 subsequently discontinued treatment. Eligible LEPR Hets patients were rare and hard to recruit Eight (8) of 11 setmelanotide-assigned patients were down classified (predominantly one variant) Positive response in 3 setmelanotide-assigned patients who had a pathogenic variant achieved BMI reductions of: -4.3%, -7.2%*, -13.3%

® 20 SRC1 (NCOA1)

® 21 SRC1 (NCOA1): Setmelanotide Achieved Statistically Significant BMI Reduction in mITT Patients Based on LOCF Analysis Post hoc analysis of mITT cohort based on last observation carried forward (LOCF) Setmelanotide (N=36) Placebo (N=37) -4.87 (1.192)1.44 (0.837)% change BMI baseline to week 52: Mean (SE) -4.51 (1.68) [-6.84, -2.18] 1.73 (1.115) [-0.50, +3.95] ANCOVA LSM (SE) [95% CI] -6.24 (1.499) [-9.33, -3.25] LSM difference (SE) [95% CI], setmelanotide-placebo <0.0001P-Value for LSM difference mITT, modified intent-to-treat
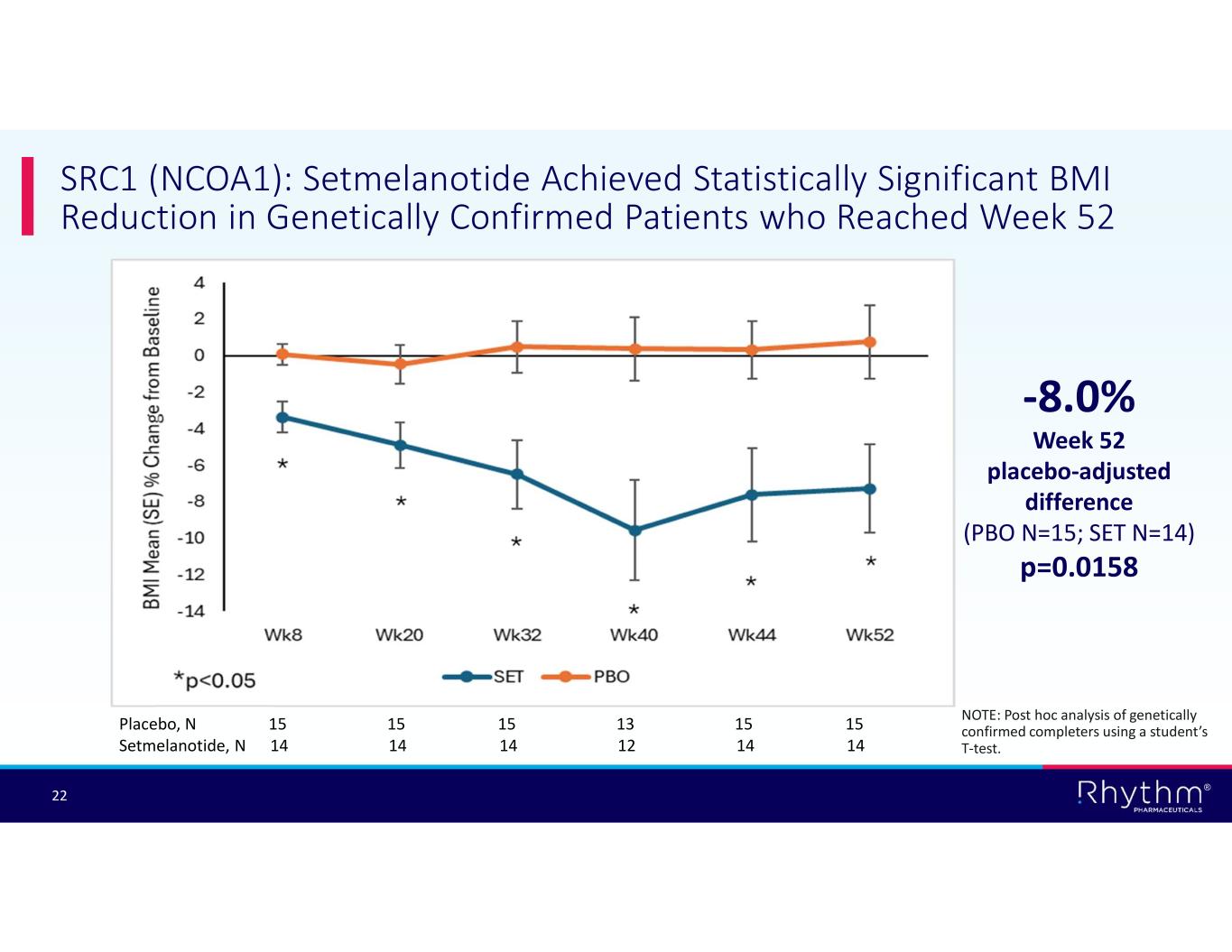
® 22 SRC1 (NCOA1): Setmelanotide Achieved Statistically Significant BMI Reduction in Genetically Confirmed Patients who Reached Week 52 -8.0% Week 52 placebo-adjusted difference (PBO N=15; SET N=14) p=0.0158 Placebo, N 15 15 15 13 15 15 Setmelanotide, N 14 14 14 12 14 14 NOTE: Post hoc analysis of genetically confirmed completers using a student’s T-test.
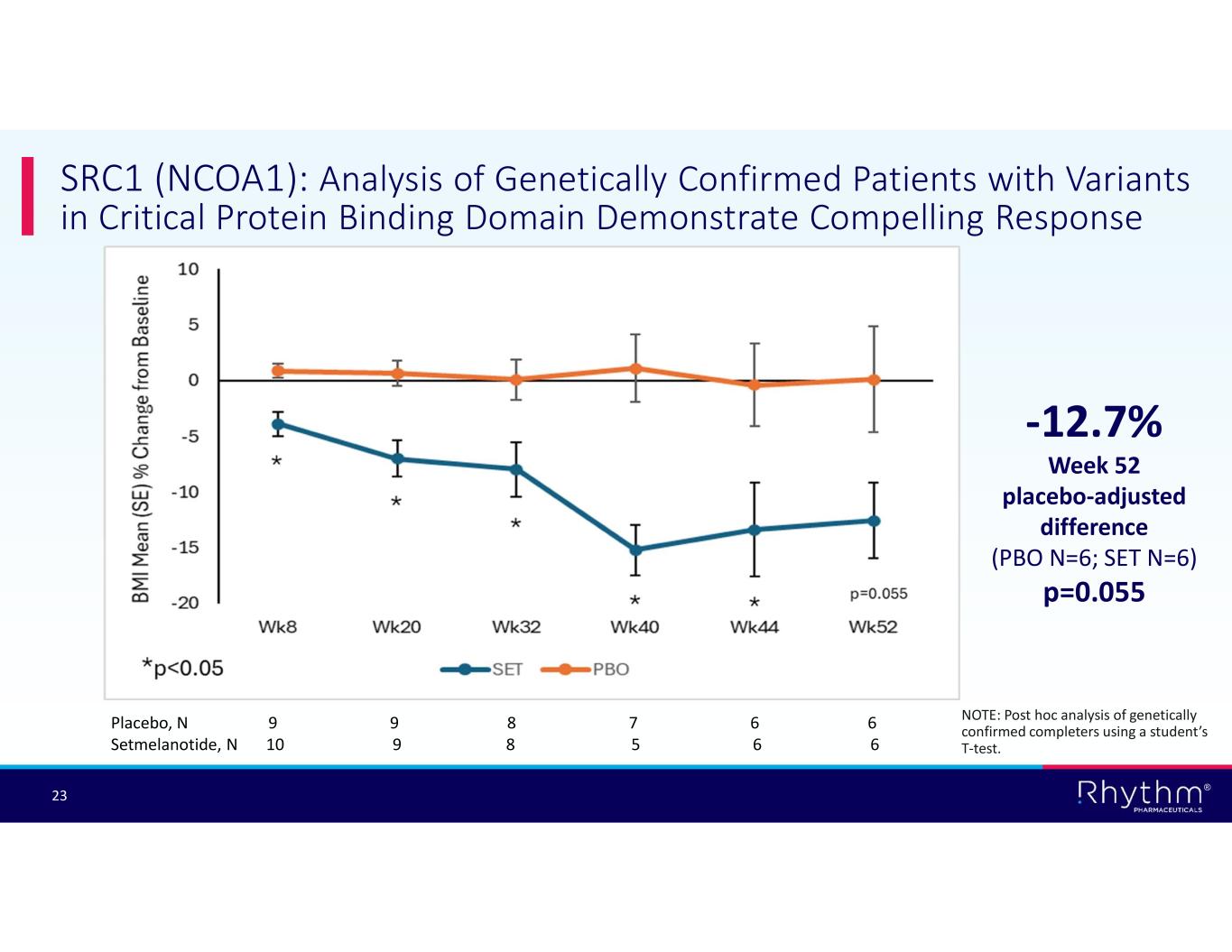
® 23 SRC1 (NCOA1): Analysis of Genetically Confirmed Patients with Variants in Critical Protein Binding Domain Demonstrate Compelling Response -12.7% Week 52 placebo-adjusted difference (PBO N=6; SET N=6) p=0.055 Placebo, N 9 9 8 7 6 6 Setmelanotide, N 10 9 8 5 6 6 NOTE: Post hoc analysis of genetically confirmed completers using a student’s T-test.

® 24 SH2B1
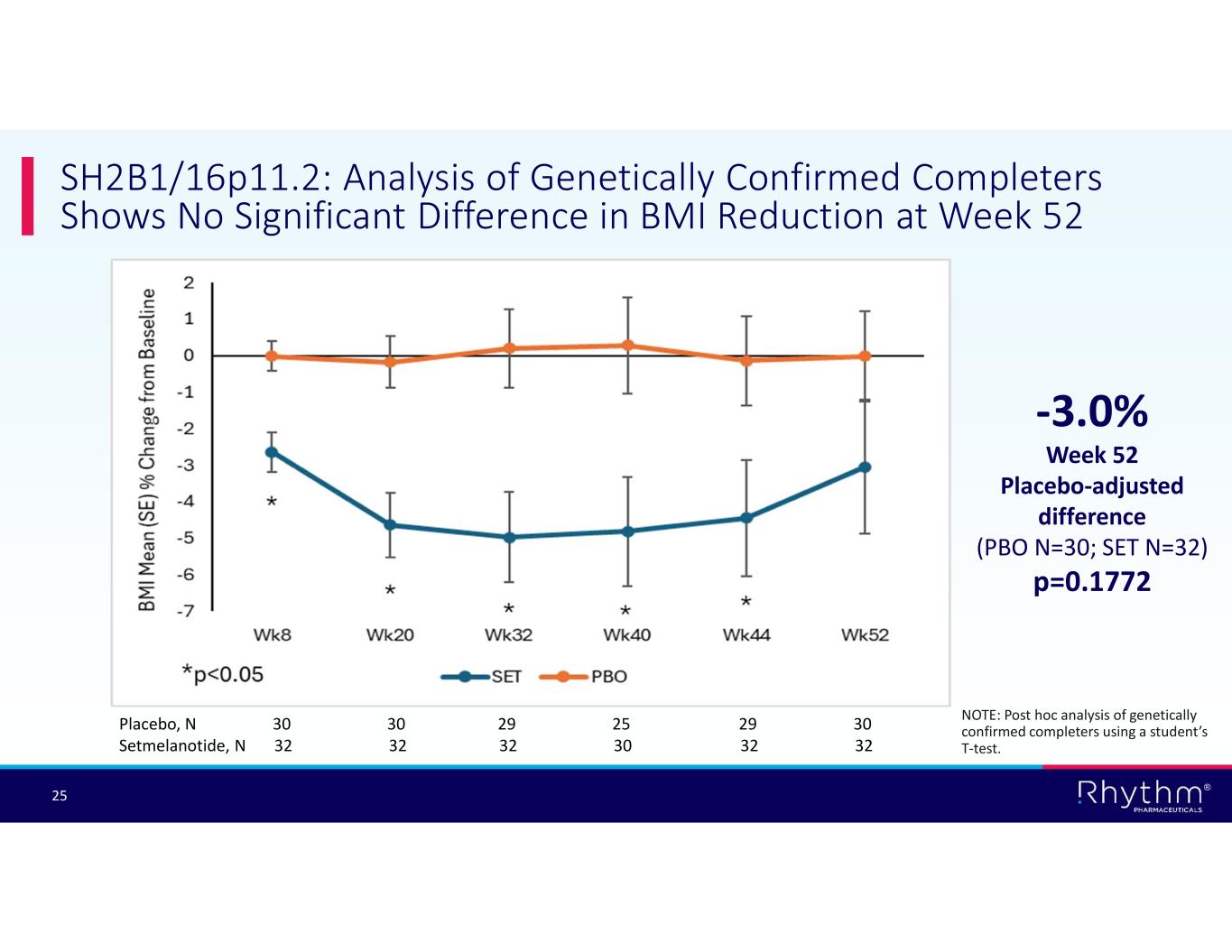
® 25 SH2B1/16p11.2: Analysis of Genetically Confirmed Completers Shows No Significant Difference in BMI Reduction at Week 52 -3.0% Week 52 Placebo-adjusted difference (PBO N=30; SET N=32) p=0.1772 Placebo, N 30 30 29 25 29 30 Setmelanotide, N 32 32 32 30 32 32 NOTE: Post hoc analysis of genetically confirmed completers using a student’s T-test.
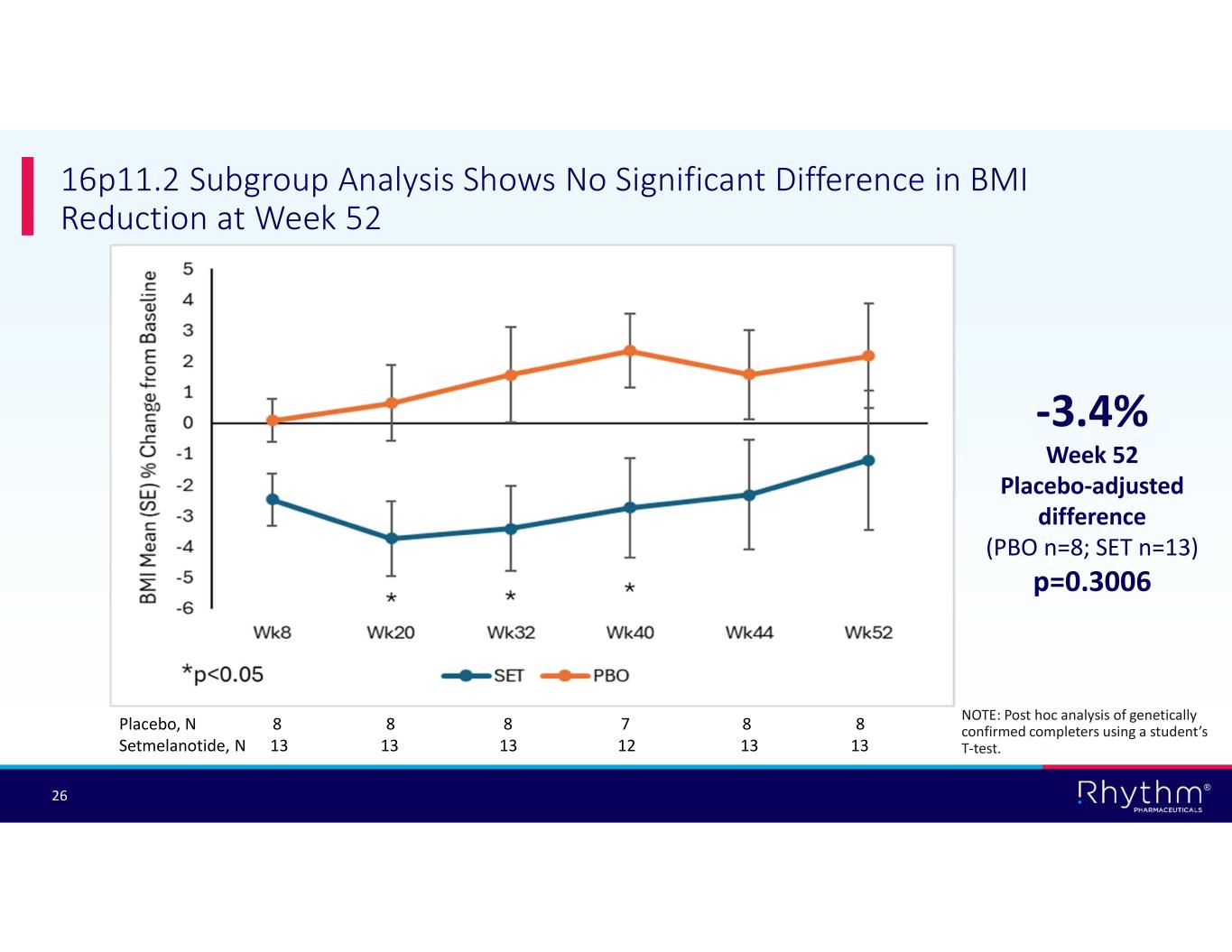
® 26 16p11.2 Subgroup Analysis Shows No Significant Difference in BMI Reduction at Week 52 Placebo, N 8 8 8 7 8 8 Setmelanotide, N 13 13 13 12 13 13 -3.4% Week 52 Placebo-adjusted difference (PBO n=8; SET n=13) p=0.3006 NOTE: Post hoc analysis of genetically confirmed completers using a student’s T-test.
® 27 Encouraging Results Leading to Next Steps Encouraged by positive analyses of POMC/PCSK1 and SRC1 substudies Continue to interrogate POMC/PCSKI, SRC1 plus DAYBREAK genes to improve LOF determination Advance next- generation MC4R agonists in genetic indications LOF, loss of function

® 28 MC4R Agonism Development Across Three Pillars Hypothalamic Obesity • U.S. launch* • EMA authorization** • Ongoing Ph2 trial with RM-718 • Ph3 trial with bivamelagon by YE 2026 Prader-Willi Syndrome • Ongoing Ph2 trial with setmelanotide • Ph1/2 trial with RM-718 initiated Genetic MC4R Pathway Diseases • Promising signals in POMC Hets and SRC1 • Additional DAYBREAK genes • Next-generation assets *Pending FDA approval; the FDA’s assigned PDUFA goal date is March 20, 2026; **pending regulatory feedback

® 29 Questions
